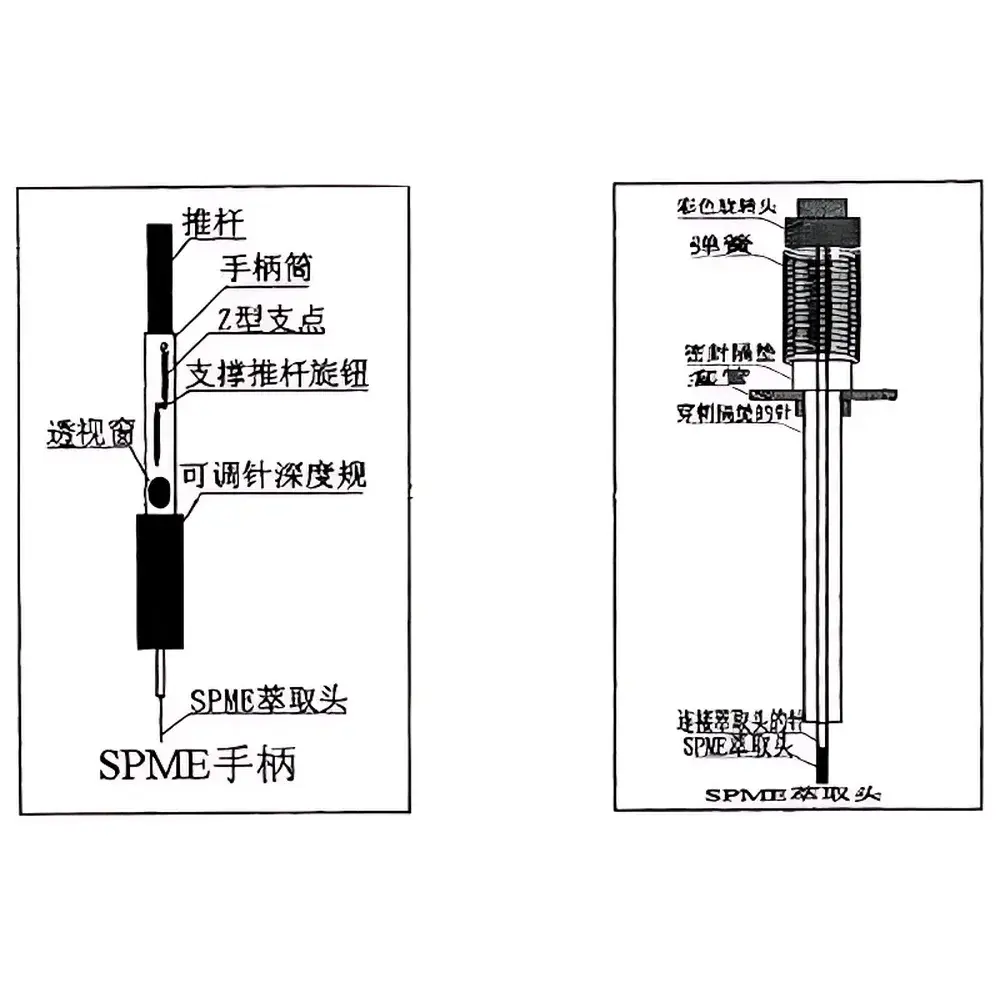
Supelco SPME Manual Solid Phase Microextraction Device
| Origin | USA |
|---|---|
| Supplier Type | Authorized Distributor |
| Import Status | Imported |
| Operation Mode | Manual |
| Price | USD 4,200 (FOB) |
Overview
The Supelco SPME Manual Solid Phase Microextraction Device is a field-deployable, syringe-style sample preparation tool engineered for equilibrium-based, solvent-free extraction of volatile and semi-volatile organic compounds from gaseous, liquid, and solid matrices. Based on the patented Solid Phase Microextraction (SPME) principle developed by Supelco (Sigma-Aldrich), this device operates via passive adsorption or absorption of analytes onto a 1 cm fused-silica fiber coated with a chemically defined stationary phase—encased within a stainless-steel sheath and mounted on a reusable manual holder. Unlike conventional liquid–liquid or solid-phase extraction methods, SPME integrates sampling, extraction, concentration, and direct instrumental introduction into a single step—eliminating solvent consumption, reducing contamination risk, and minimizing procedural variability. Its design conforms to fundamental chromatographic partitioning theory: analyte distribution between sample matrix and coating phase follows Nernst partitioning behavior, enabling quantitative and reproducible enrichment without calibration drift from matrix effects. The device is fully compatible with GC, GC–MS, LC, and LC–MS platforms, supporting both headspace and direct-immersion modes per ASTM D7622–21 and ISO 17935:2015 guidelines for environmental and forensic trace analysis.
Key Features
- Reusable stainless-steel manual holder (Cat. No. 57330U) with ergonomic thumbwheel actuation for precise fiber extension/retraction
- 1 cm fused-silica fiber coated with application-optimized stationary phases—including PDMS (non-polar), PA (polar), PDMS/DVB (mixed-mode), CAR/PDMS (for VOCs), CW/DVB (for alcohols), and CW/TPR (for surfactants)
- Fiber diameters ranging from 7 µm to 100 µm, selected based on analyte volatility (MW 30–600 Da) and polarity—enabling method-specific optimization per USP and EPA Method 8261B
- No solvent required; no derivatization or centrifugation needed—reducing total sample prep time to under 15 minutes
- Robust architecture with thermal stability up to 250 °C (fiber-dependent); compatible with standard GC inlet liners and autosampler vial septa
- Designed for GLP-compliant workflows: fiber batch certification, lot-specific performance data, and traceable manufacturing per ISO 9001:2015
Sample Compatibility & Compliance
The SPME manual system supports diverse sample types across regulated and research environments: aqueous environmental samples (drinking water, wastewater, leachates), biological fluids (urine, plasma, saliva), air/headspace vapors, soil/sediment headspace, food volatiles, and forensic evidence (arson residues, drug metabolites). It meets critical compliance requirements for method validation under FDA 21 CFR Part 11 (when paired with compliant GC/LC software), EU Directive 2009/128/EC (pesticide residue analysis), and CLSI EP29-A3 for precision assessment. Each fiber type undergoes rigorous QC testing for coating thickness uniformity (±5% CV), breakthrough volume (≥100 ng for benzene on 75 µm CAR/PDMS), and carryover (<0.1% after 250 °C bakeout). Full documentation—including Certificate of Analysis, Coating Stability Report, and ISO/IEC 17025-accredited test summaries—is provided with every shipment.
Software & Data Management
While the manual SPME device operates independently of software, its integration into validated analytical workflows requires traceable data handling. When coupled with GC or LC systems, SPME-derived chromatograms are processed using vendor-neutral software (e.g., OpenLab CDS, Chromeleon, or MassHunter) supporting audit trails, electronic signatures, and metadata tagging per FDA 21 CFR Part 11 Annex 11. Fiber usage logs, exposure times, desorption temperatures, and matrix-matched calibration curves are stored as structured metadata. For laboratories implementing LIMS, SPME fiber lot numbers, expiration dates, and reuse cycles (up to 100 extractions for PDMS fibers under controlled conditions) can be mapped to sample IDs—ensuring full chain-of-custody for forensic and clinical applications.
Applications
This SPME platform is routinely deployed in: environmental monitoring (EPA Method 8261B for VOCs in water), clinical toxicology (detection of amphetamines and benzodiazepines in urine), forensic chemistry (accelerant identification in arson investigations), pharmaceutical QC (residual solvent analysis per ICH Q2(R2)), flavor & fragrance profiling (C3–C20 carbonyls and esters), and defense-related chemical threat screening (nerve agent hydrolysis products in wipe samples). Its portability enables on-site sampling in wastewater treatment plants, crime scenes, and field epidemiology studies—where immediate stabilization of labile analytes (e.g., aldehydes, thiols) is critical to data integrity.
FAQ
What is the maximum recommended number of extractions per SPME fiber?
PDMS and PA fibers are validated for up to 100 extractions under optimized thermal desorption conditions; CAR/PDMS and PDMS/DVB fibers are rated for 50–75 uses depending on matrix cleanliness and desorption temperature.
Can SPME fibers be used with HPLC systems?
Yes—fibers with CW/TPR, PDMS/DVB (60 µm), and PA coatings are specifically designed for LC-compatible desorption using solvent elution (e.g., acetonitrile/methanol) prior to injection; dedicated holders (Cat. No. 57347U) support automated LC integration.
How should SPME fibers be conditioned before first use?
All fibers require pre-conditioning at their upper thermal limit (e.g., 250 °C for PDMS, 230 °C for PA) for 30 minutes in a GC inlet or dedicated oven to remove residual solvents and stabilize coating morphology.
Is SPME suitable for quantitative analysis?
Yes—when combined with internal standards (e.g., deuterated analogs) and matrix-matched calibration, SPME delivers RSDs <5% for repeatability and recovery rates of 85–115% across validated methods per ICH Q2(R2) guidance.
What safety certifications apply to the manual SPME holder?
The holder complies with CE marking (2014/30/EU EMC Directive), RoHS 2011/65/EU, and UL 61010-1 for laboratory electrical equipment—though no power source is integrated, its mechanical design meets ISO 13850 emergency stop compatibility standards for shared instrumentation workspaces.