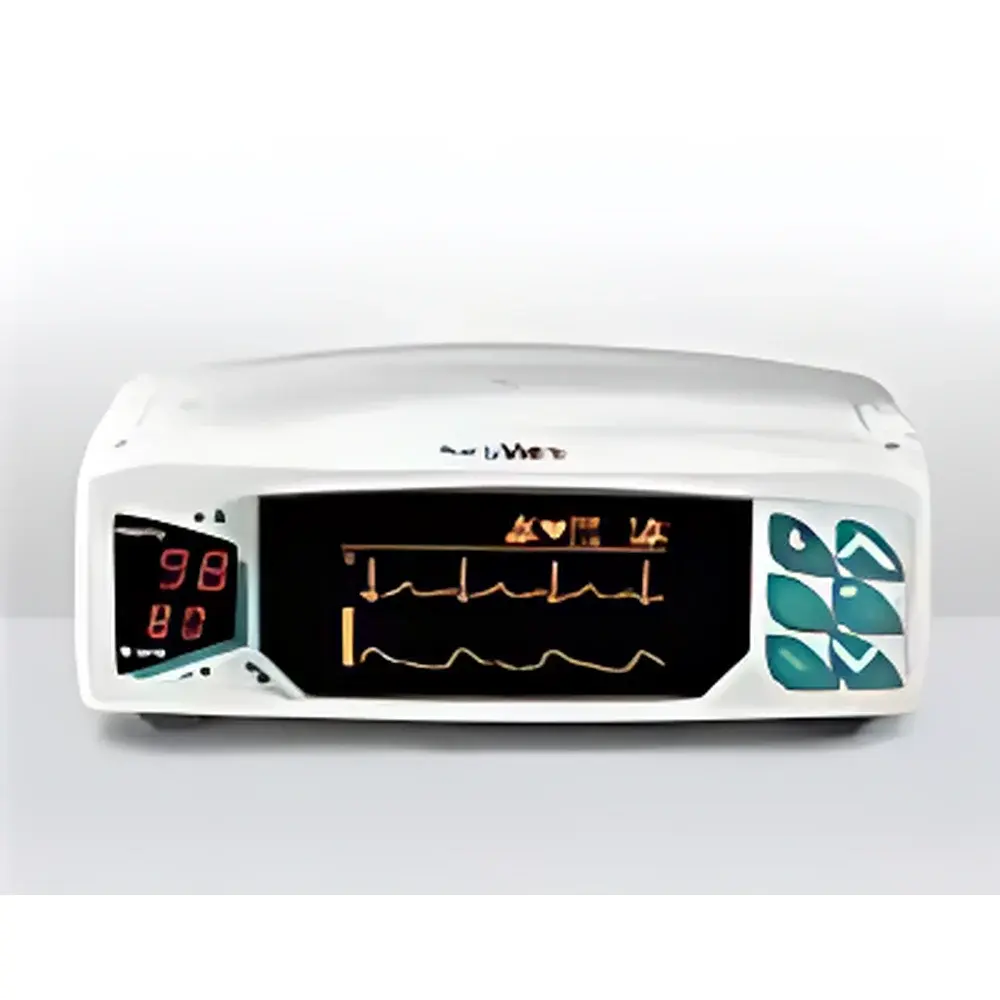
SurgiVet V3404 Veterinary Electrocardiography Monitor
| Brand | Harvard Apparatus |
|---|---|
| Origin | USA |
| Model | V3404 |
| ECG Leads | 3- or 5-lead configuration |
| Heart Rate Range | 15–350 bpm |
| Display | Backlit LCD |
| Power | Rechargeable internal battery (optional AC adapter) |
| Connectivity | RS-232 serial interface (V3362 cable optional) |
| Printer Option | V3408 thermal printer module |
| Parameters Measured | ECG waveform, SpO₂, Temperature (rectal/probe), Respiration rate (impedance-based) |
Overview
The SurgiVet V3404 Veterinary Electrocardiography Monitor is a dedicated, compact, and clinically validated physiological monitoring system engineered for real-time, multi-parameter assessment of small to medium-sized laboratory and companion animals during surgical procedures, recovery, and longitudinal studies. Built upon standardized electrocardiographic principles—specifically bipolar and augmented unipolar limb lead configurations—the V3404 acquires, filters, and displays high-fidelity ECG waveforms in accordance with ANSI/AAMI EC13:2002 and IEC 60601-2-27 requirements for veterinary electrocardiographs. Its primary design objective is to support rigorous cardiovascular assessment in controlled environments including AAALAC-accredited vivaria, GLP-compliant preclinical research labs, and clinical veterinary operating rooms. The device operates as a standalone unit with integrated signal conditioning circuitry optimized for low-amplitude, high-noise biological signals typical of rodent, rabbit, canine, and feline models.
Key Features
- Configurable 3-lead or 5-lead ECG acquisition with selectable filter settings (e.g., 0.5–40 Hz diagnostic mode; 0.5–150 Hz surgical mode) to suppress motion artifact and electromagnetic interference
- Heart rate calculation algorithm compliant with AHA/ACC guidelines, reporting instantaneous and averaged values across 15-, 30-, and 60-second windows
- Reflectance-based pulse oximetry (SpO₂) with dual-wavelength (660 nm / 940 nm) LED sensor, calibrated for hemoglobin saturation estimation in species-specific perfusion ranges
- Digital temperature measurement via precision thermistor probe (±0.1°C accuracy over 20–45°C range), compatible with rectal, esophageal, or surface mounting
- Impedance pneumography for non-invasive respiration rate detection, with adjustable sensitivity thresholds to accommodate species-dependent tidal volume profiles
- Backlit monochrome LCD display (128 × 64 pixels) with configurable waveform scaling, timebase (12.5–50 mm/s), and on-screen annotation markers
- Rechargeable lithium-ion battery supporting ≥4 hours of continuous operation; battery status indicator and low-power warning per ISO 80601-2-56
- Programmable alarm limits with visual (flashing icons) and audible (adjustable tone/pitch) alerts for arrhythmia detection, SpO₂ desaturation, bradycardia/tachycardia, and probe disconnection
Sample Compatibility & Compliance
The V3404 is validated for use across common preclinical species including mice, rats, guinea pigs, rabbits, dogs, cats, and non-human primates. Electrode placement follows standardized veterinary lead systems (e.g., Lead I, II, III, aVR, aVL, aVF) and accommodates both adhesive disposable electrodes and reusable stainless-steel needle electrodes. All transducers and cables meet IEC 60601-1 third edition safety requirements for applied parts and electrical isolation. The monitor complies with FDA 21 CFR Part 11 when used with validated data export workflows and supports audit trail generation when paired with compatible software platforms. It is suitable for use in facilities maintaining AAALAC International accreditation and adhering to NIH Guide for the Care and Use of Laboratory Animals standards.
Software & Data Management
Raw analog outputs (ECG, SpO₂, temperature, respiration) are accessible via BNC connectors for integration with external DAQ systems (e.g., LabChart, AcqKnowledge). Optional RS-232 serial communication (V3362 interface cable) enables ASCII-formatted data streaming at 9600 baud to host PCs running custom or third-party acquisition software. While the V3404 does not include embedded storage, timestamped parameter logs can be captured externally with software supporting serial protocol parsing. For regulatory documentation, exported datasets may be structured to align with CDISC SDTM conventions for cardiovascular endpoints. The V3408 thermal printer provides hard-copy output of trend graphs and snapshot waveforms, meeting traceability requirements under GLP Annex 11 and ISO/IEC 17025.
Applications
- Intraoperative cardiovascular monitoring during terminal or survival surgeries in rodents and larger mammals
- Pharmacodynamic evaluation of cardioactive compounds (e.g., antiarrhythmics, beta-blockers, ion channel modulators)
- Longitudinal telemetry correlation studies where spot-check validation of implanted device readings is required
- Teaching laboratories for hands-on instruction in comparative electrophysiology and anesthetic depth assessment
- Post-anesthesia recovery monitoring in veterinary clinical settings with documented SOPs
- ISO 10993 biocompatibility testing where physiologic stability must be confirmed during material exposure
FAQ
Is the V3404 compatible with rodent ECG measurements?
Yes—the device’s high-input impedance (>10 MΩ), low-noise amplification stage, and adjustable gain (1–5 mV/div) enable reliable R-wave detection in mice and rats when used with appropriate needle electrodes and proper grounding.
Does the V3404 support FDA 21 CFR Part 11 compliance?
The hardware itself does not include electronic signature or audit trail functionality; however, when integrated into a validated system using compliant software (e.g., LabChart with Part 11 add-on), it meets data integrity requirements for regulated studies.
Can the V3404 store data internally?
No—it is a real-time display and alarm device without onboard memory; continuous recording requires external PC-based acquisition software.
What is the recommended recalibration interval?
Harvard Apparatus recommends annual verification against NIST-traceable test signals per manufacturer’s service manual, with functional checks performed before each experimental session.
Is the V3404 certified for use in explosion-proof environments?
No—it is not intrinsically safe or rated for hazardous locations; use is restricted to standard laboratory and clinical spaces meeting IEC 60601-1 environmental class I, IP20 specifications.