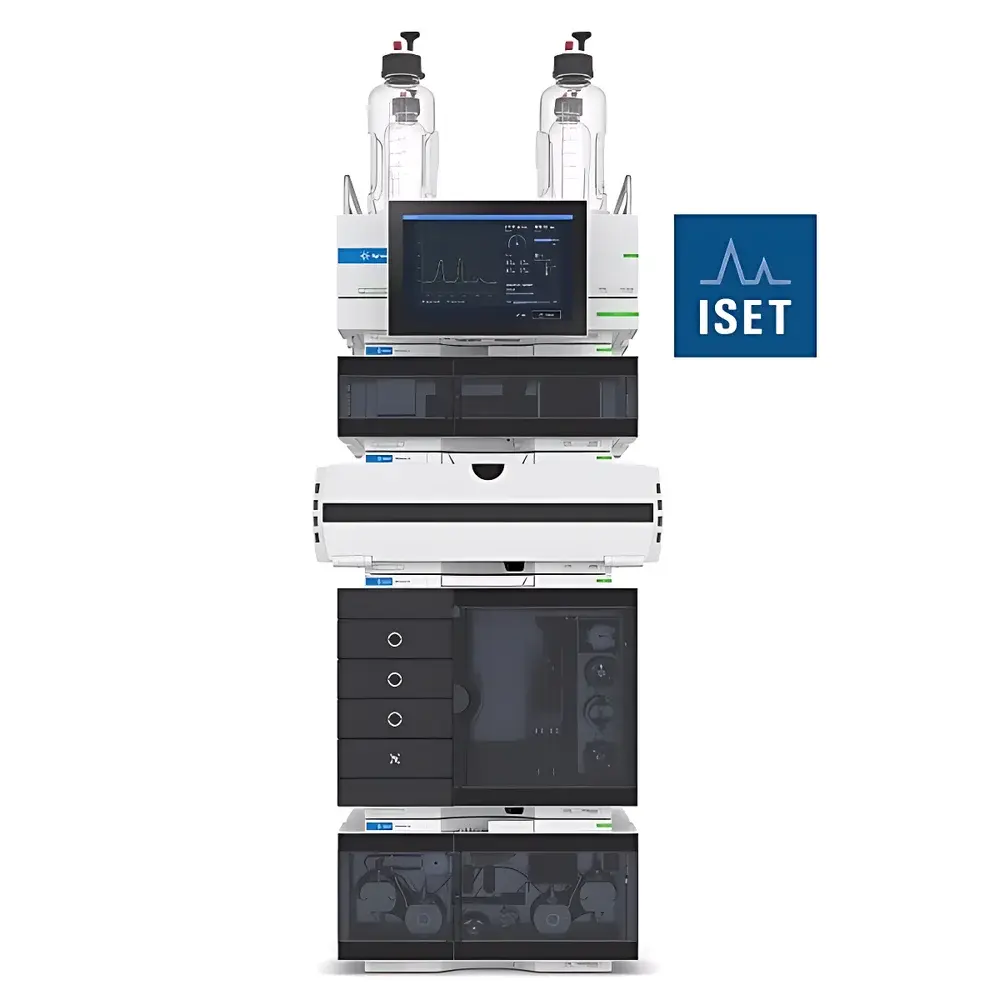
Agilent 1290 Infinity III Method Transfer System
| Brand | Agilent Technologies |
|---|---|
| Origin | Germany |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Origin Category | Imported |
| Model | 1290 Infinity III Method Transfer |
| Instrument Type | Conventional and Ultra-High-Performance Liquid Chromatograph (HPLC/UHPLC) |
| Application Scope | General-purpose chromatographic analysis |
| Flow Rate Range | Up to 5 mL/min (with G7120A and G7104A pumps) |
| Maximum Operating Pressure | 1300 bar |
| Injection Volume Range | 0.1–100 µL (expandable to 0.1–900 µL with optional extended range kit |
| Temperature Control | Dual-zone capability (G7116A/B thermostatted compartments) |
| Solvent Selection | Up to 4 solvents standard |
| Column ID Reader | Optional |
| Column Capacity | 8 positions |
| Dimensions (W × D) | 435 mm × 468 mm |
| Power Supply | 100–240 VAC |
Overview
The Agilent 1290 Infinity III Method Transfer System is a purpose-built, high-fidelity liquid chromatography platform engineered to address the critical challenge of cross-platform method portability in regulated and research laboratories. Leveraging Intelligent System Emulation Technology (ISET), this system mathematically models the dwell volume, gradient delay, dispersion characteristics, and pump/compression dynamics of legacy or third-party HPLC and UHPLC instruments—enabling precise replication of retention times, peak shape, resolution, and selectivity without physical re-optimization. Unlike conventional method transfer workflows that rely on empirical column scaling or manual parameter adjustments, ISET operates on first-principles chromatographic modeling grounded in system dwell time characterization, compressibility-corrected flow delivery, and thermally stabilized detection pathways. Designed for laboratories managing heterogeneous instrument fleets—including Agilent, Waters, Shimadzu, and Thermo Fisher systems—the 1290 Infinity III Method Transfer System serves as a centralized, auditable reference platform compliant with ICH Q5A, Q5C, and FDA guidance on analytical method transfer.
Key Features
- Intelligent System Emulation Technology (ISET): Fully automated emulation of >30 commercially available HPLC/UHPLC systems—including dwell volume, gradient delay, and dispersion profiles—via built-in instrument library and user-defined system characterization.
- Ultra-High-Pressure Capability: 1300 bar maximum operating pressure supports sub-2 µm particle columns and rapid gradient separations while maintaining baseline stability under high-backpressure conditions.
- Dual-Zone Thermal Management: Independent temperature control of autosampler (G7116A) and column compartment (G7116B) ensures reproducible retention across ambient fluctuations and enables method robustness testing per ICH Q2(R2).
- Extended Injection Flexibility: Standard 0.1–100 µL range; expandable to 0.1–900 µL (extended loop kit) or 0.1–1500 µL (multi-draw toolkit), supporting trace analysis, preparative-scale injections, and dilution series without hardware swaps.
- Modular Solvent Delivery: Four-solvent quaternary pumping standard; scalable to 15 solvents via optional Solvent Selection Valve—ideal for complex gradient methods requiring orthogonal mobile phases.
- ACT-Certified Sustainability: Independently verified by My Green Lab for environmental impact across product lifecycle (materials, energy use, end-of-life), earning the ACT Label for Accountability, Consistency, and Transparency.
Sample Compatibility & Compliance
The system accommodates standard 2.1–4.6 mm ID analytical columns (up to 8-position tray), including silica-based, hybrid, and superficially porous particles (SPP). It supports all common reversed-phase, HILIC, ion-exchange, and size-exclusion applications. From a regulatory perspective, the 1290 Infinity III Method Transfer System is validated for use in GLP and GMP environments. Its software architecture complies with 21 CFR Part 11 requirements for electronic records and signatures, including full audit trail, user access controls, and electronic signature enforcement. All ISET-generated method files retain full traceability—including source instrument model, calibration date, and emulation validation report—supporting inspection readiness per EMA and PMDA expectations.
Software & Data Management
Controlled via OpenLab CDS 2.x or MassHunter Quantitative Analysis software, the system integrates seamlessly into enterprise-wide data management frameworks. ISET simulation reports include overlay chromatograms, overlay metrics (ΔtR, %RSD of resolution), and uncertainty propagation analysis for each transferred parameter. Raw data are stored in vendor-neutral .d format, compatible with third-party processing tools (e.g., Chromeleon, ACD/Labs). All method transfer activities—including system characterization runs, emulation validation, and final report generation—are captured in an immutable audit trail with timestamped operator identity and reason-for-change fields. Optional integration with Agilent’s Enterprise Informatics suite enables automatic synchronization with LIMS and ELN systems.
Applications
- Regulatory method transfer between QC labs using disparate instrument platforms (e.g., from Waters Acquity UPLC to Agilent 1290 Infinity III).
- Contract manufacturing organization (CMO) support: Reproducing client-developed methods on-site without method revalidation.
- Method lifecycle management: Updating legacy HPLC methods to UHPLC-ready formats while preserving pharmacopeial compliance (USP , EP 2.2.46).
- Training harmonization: Standardizing analyst training on a single platform instead of multiple vendor-specific instruments.
- Green chemistry initiatives: Reducing solvent consumption and instrument footprint through consolidation of legacy systems into one high-efficiency platform.
FAQ
How does ISET ensure retention time accuracy during method transfer?
ISET performs real-time correction of dwell volume differences and gradient delay effects using empirically measured system parameters—not generic assumptions—resulting in typical ΔtR ≤ ±0.05 min across 95% of small-molecule methods.
Can ISET emulate non-Agilent detectors (e.g., photodiode array or MS interfaces)?
ISET emulates the LC system only (pump, injector, column oven, dwell path); detector response is preserved as acquired. For MS-coupled transfers, signal intensity normalization is handled externally via acquisition software alignment algorithms.
Is method revalidation required after ISET-based transfer?
Per ICH Q2(R2), partial revalidation (system suitability, specificity, precision) is recommended; full revalidation is typically unnecessary if the receiving system’s performance qualification meets original method specifications.
What documentation is generated to support regulatory submissions?
Each ISET session exports a PDF validation report containing source/target system definitions, overlay chromatograms, resolution/tailing comparisons, and a signed declaration of equivalence per ICH Q5C Annex 2.
Does the dual-zone temperature control support column switching or heart-cutting applications?
Yes—G7116A/B modules support independent setpoints and ramp profiles, enabling synchronized thermal programming for comprehensive 2D-LC configurations when paired with Agilent’s 2D-LC solution kits.