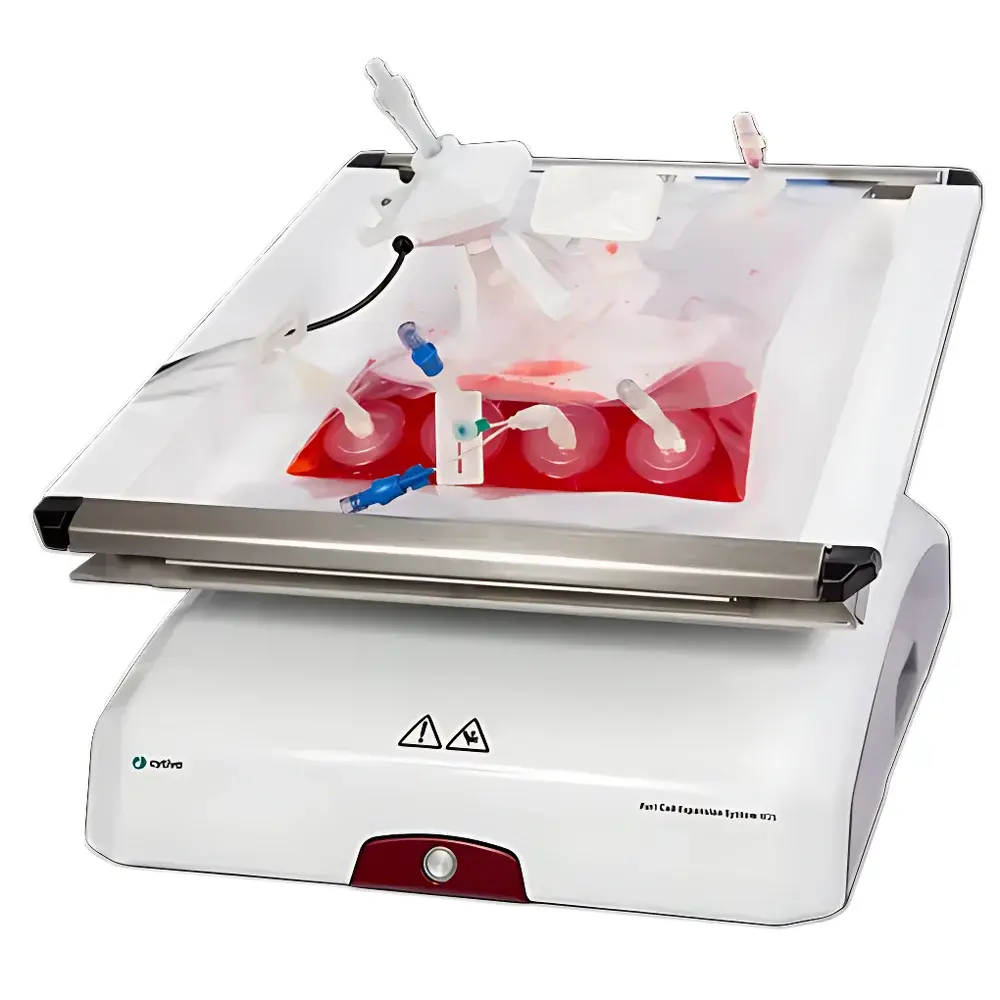
Cytiva Xuri Cell Expansion System W25
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Xuri Cell Expansion System W25 |
| Price | Upon Request |
| Temperature Range | 15°C–32°C |
| Working Volume | 25 L |
Overview
The Cytiva Xuri Cell Expansion System W25 is an automated, single-use, rocking-motion bioreactor platform engineered for scalable suspension cell culture in cell therapy research, process development, and clinical- or commercial-scale manufacturing. It operates on the well-established WAVE™ rocking technology—a gravity-driven, low-shear mixing principle that generates gentle wave motion within a sterile, disposable BioBag™ bioprocess container. This hydrodynamic environment ensures uniform nutrient distribution, efficient gas exchange (O₂/CO₂), and minimal mechanical stress on sensitive mammalian cells—critical for maintaining viability, phenotype stability, and functional potency during expansion. Designed for operation in regulated environments, the W25 supports working volumes up to 25 L in a single-use bag, eliminating open transfers, reducing operator intervention, and minimizing contamination risk. Its closed architecture complies with fundamental principles of aseptic processing and aligns with ICH Q5A(R2) and Q5D guidance on cell line characterization and culture system validation.
Key Features
- Gentle, low-shear rocking motion (0.5–30° tilt angle; 5–30 rpm range) optimized for high-density suspension cultures of T cells, NK cells, CAR-T cells, and other therapeutic primary or immortalized lines.
- Integrated environmental control: precise regulation of temperature (15–32°C), dissolved oxygen (DO), pH (via optional CO₂/gas blending module), and perfusion rate using peristaltic pumps.
- UNICORN® 7 software platform with full audit trail, electronic signatures, and 21 CFR Part 11 compliance—including user access levels, change control logs, and secure data archiving.
- Real-time remote monitoring and control via Ethernet/Wi-Fi connectivity; configurable alarm thresholds for critical parameters (e.g., DO deviation >±5%, temperature drift >±0.5°C, bag volume limits).
- Pre-qualified, gamma-irradiated BioBag™ W25 containers (25 L max fill volume) with integrated sensors (optional DO/pH), sterile connectors, and single-use fluid paths—validated for extractables/leachables per USP and ISO 10993-18.
- Compact footprint (W × D × H: 720 × 740 × 1050 mm) suitable for Class A/B cleanroom integration or GMP-compliant laboratory spaces.
Sample Compatibility & Compliance
The Xuri W25 accommodates a broad range of suspension-adapted mammalian cell types, including but not limited to human peripheral blood mononuclear cells (PBMCs), CD34⁺ hematopoietic stem cells, hybridomas, and recombinant CHO or HEK293 lines used in viral vector production. All wetted components are manufactured from USP Class VI-certified materials and undergo rigorous biocompatibility testing. The system supports adherence to current Good Manufacturing Practice (cGMP), EU Annex 1 (2022), and FDA’s Process Validation Guidance (2011). IQ/OQ documentation packages—including installation verification, sensor calibration records, and operational performance qualification protocols—are available upon request and support regulatory submissions under IND/IMPD pathways.
Software & Data Management
UNICORN® 7 serves as the embedded control and data acquisition engine. It enables creation, versioning, and execution of multi-step perfusion protocols with time- or event-triggered actions (e.g., feed initiation at viable cell density >5 × 10⁶ cells/mL). All raw sensor data (temperature, DO, pH, rocking speed, pump RPM) are timestamped, encrypted, and stored locally with automatic backup to network drives. Audit trails meet ALCOA+ criteria (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) and are exportable in CSV/PDF formats for quality review or regulatory inspection. Optional integration with MES or LIMS systems is supported via OPC UA or RESTful API interfaces.
Applications
- Clinical-grade expansion of autologous and allogeneic T/NK cells for Phase I–III trials.
- Process development and scale-down modeling for upstream bioprocessing of lentiviral or retroviral vectors.
- Seed train generation for larger stainless-steel or single-use bioreactors (e.g., transition from W25 to 200 L or 500 L platforms).
- Feasibility studies of novel media formulations, feeding strategies, or metabolic modulators under controlled DO/pH setpoints.
- Stability assessment of cell products across multiple passages under GMP-like conditions.
FAQ
Is the Xuri W25 compliant with 21 CFR Part 11?
Yes—the UNICORN® 7 software includes role-based access control, electronic signatures, audit trail logging, and data integrity safeguards validated per GAMP 5 and aligned with FDA expectations.
Can the system be integrated into an existing automation infrastructure?
Yes—via OPC UA server functionality or custom REST API endpoints for bidirectional communication with SCADA, MES, or enterprise quality systems.
What validation support is provided for GMP use?
Cytiva offers comprehensive IQ/OQ documentation templates, pre-executed test scripts, and on-site qualification support by certified field service engineers.
Are BioBag™ containers supplied sterile and ready-to-use?
Yes—all W25 bags are gamma-irradiated (25–45 kGy), certified sterile (SAL ≤10⁻⁶), and shipped in double-barrier packaging with full CoA and CoC documentation.
Does the system support pH control without external gas blending?
Basic pH monitoring is available; active pH control requires optional integrated CO₂/N₂ gas mixer and mass flow controllers, configured during system specification.