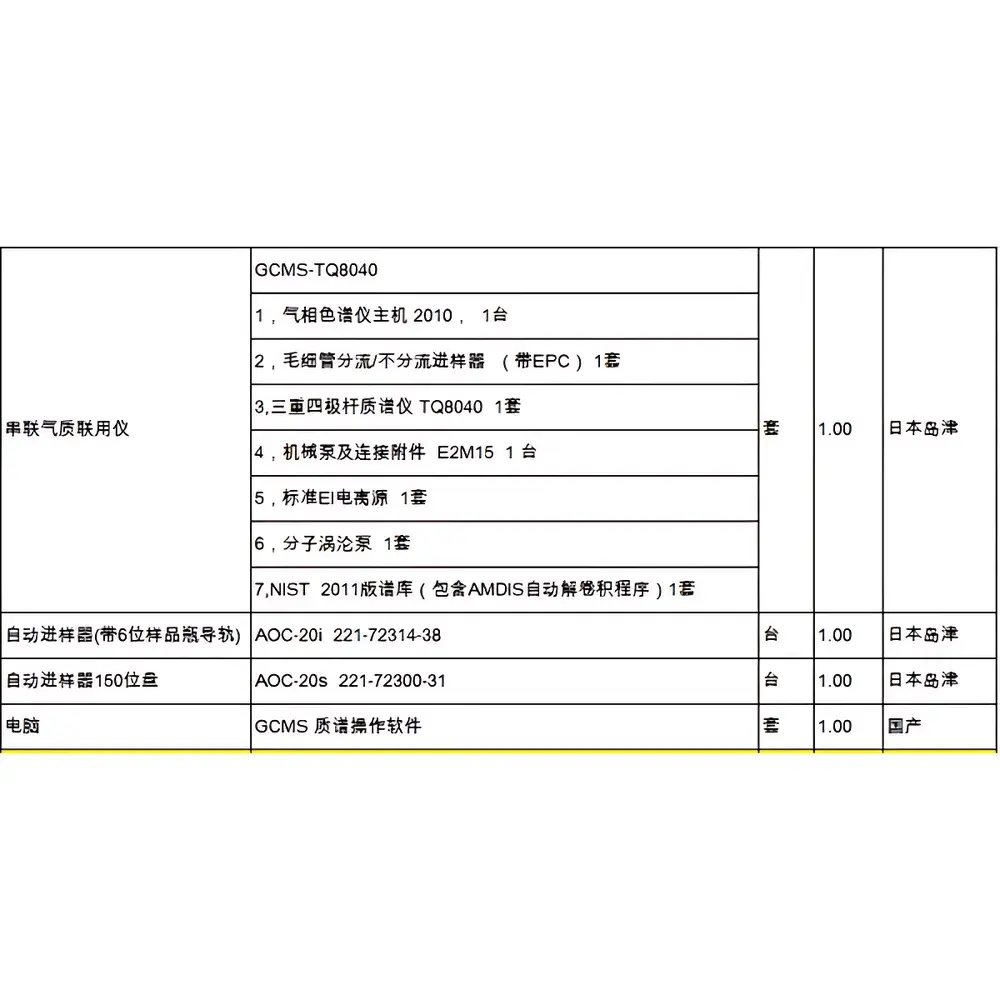
Shimadzu GCMS-TQ8040 Triple Quadrupole Gas Chromatography-Mass Spectrometer (Refurbished)
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | GCMS-TQ8040 |
| Configuration | Refurbished Unit with Full Functional Validation |
| Compliance | Meets Original Shimadzu Factory Specifications for GC-MS/MS Operation |
| Software | Includes Licensed GCMSsolution Ver. 5.x (with Smart MRM, ASSP, and Scan/MRM Simultaneous Acquisition Modules) |
| Instrument Status | Fully Recertified with Performance Verification Report (PVR) per ISO/IEC 17025 Traceable Protocols |
Overview
The Shimadzu GCMS-TQ8040 is a triple quadrupole gas chromatography–tandem mass spectrometry (GC-MS/MS) system engineered for high-sensitivity, high-selectivity quantitative analysis of trace-level organic compounds in complex matrices. Operating on the principle of selective reaction monitoring (SRM) within a tandem mass spectrometric architecture—where Q1 isolates precursor ions, Q2 performs collision-induced dissociation (CID), and Q3 detects characteristic product ions—the GCMS-TQ8040 delivers exceptional specificity and robustness for regulated environments. Designed and originally manufactured in Kyoto, Japan, this refurbished unit retains full hardware integrity and undergoes comprehensive functional recertification, including ion source cleaning, detector gain calibration, vacuum system leak testing, and mass axis verification. Its core analytical strength lies in its ability to resolve co-eluting analytes and suppress chemical noise through MS/MS filtering—making it particularly suited for applications in environmental residue testing, food safety screening, clinical toxicology, and pharmaceutical impurity profiling.
Key Features
- Smart MRM Technology: Enables automated method setup for up to 32,768 MRM transitions per acquisition sequence—supporting large-scale multi-analyte panels without manual optimization.
- High-Luminosity Ion Source: Features an optimized electron ionization (EI) source with extended filament life and stable emission current, ensuring reproducible ion generation across extended run times.
- Advanced Scanning Speed Protocol (ASSP): Dynamically adjusts dwell time and cycle time based on peak width and transition count, preserving quantitative accuracy during ultra-fast chromatographic gradients.
- Simultaneous Scan/MRM Acquisition: Allows concurrent full-scan MS data collection (for library matching and unknown identification) alongside targeted MRM quantification—eliminating the need for separate injections.
- Collision Cell Efficiency: Utilizes a pressurized RF-only quadrupole collision cell with optimized argon or nitrogen gas flow control, delivering consistent fragmentation efficiency and low background noise.
- Robust Vacuum Architecture: Dual-stage differential pumping system with turbomolecular pump (≥300 L/s) and backing pump ensures stable operating pressure (<1 × 10⁻⁶ Pa) under continuous high-throughput conditions.
Sample Compatibility & Compliance
The GCMS-TQ8040 accepts standard 10–100 µL liquid injections via autosampler (AOC-20i or AOC-6000+ compatible), as well as headspace, purge-and-trap, and thermal desorption interfaces. It supports all common GC columns (e.g., Rtx-5MS, DB-5ms, HP-5MS; ID 0.25–0.32 mm, length 15–30 m). The instrument complies with key regulatory frameworks for analytical instrumentation, including ASTM D5845 (determination of organophosphorus pesticides), ISO 17025:2017 requirements for method validation, and USP guidelines for specificity and selectivity assessment. When operated with audit-trail-enabled GCMSsolution software (configured per FDA 21 CFR Part 11 requirements), it supports GLP and GMP-compliant workflows—including electronic signatures, user access controls, and immutable raw data archiving.
Software & Data Management
Controlled by Shimadzu’s GCMSsolution Ver. 5.x platform, the system provides integrated instrument control, real-time data acquisition, and post-run processing. Key modules include AutoReport for customizable PDF report generation, Library Search (NIST/EPA/WHO libraries), and Quant Browser for batch processing of calibration curves and QC samples. All raw data files (.qgd format) are stored in a structured directory hierarchy with embedded metadata (acquisition date, operator ID, method name, instrument serial number). Backup and restoration procedures follow ISO/IEC 27001-aligned data governance protocols. Remote diagnostics capability allows secure vendor-assisted troubleshooting without compromising network security.
Applications
- Quantitative determination of pesticide residues in fruits, vegetables, and cereals (EU MRL compliance testing)
- Screening and confirmation of mycotoxins (e.g., aflatoxin B1, ochratoxin A) in feed and grain commodities
- Residual solvent analysis in pharmaceutical active ingredients per ICH Q3C guidelines
- Endocrine-disrupting compound profiling in wastewater and surface water (EPA Method 1694)
- Forensic toxicology screening for controlled substances and metabolites in biological fluids
- Impurity identification and quantification in small-molecule drug development (ICH Q5A/Q5D)
FAQ
Is this a factory-refurbished unit or dealer-refurbished?
This GCMS-TQ8040 has undergone full functional refurbishment by an ISO 13485-certified third-party service provider authorized by Shimadzu Japan, including replacement of consumables (ion source components, detector, column nut seals), firmware update to latest stable release, and issuance of a traceable Performance Verification Report.
Does the system include original Shimadzu software licenses?
Yes—this unit ships with a fully licensed copy of GCMSsolution Ver. 5.61 (or later), pre-installed and activated with perpetual usage rights for the registered instrument serial number.
What documentation accompanies the instrument?
You will receive the original Shimadzu operation manual (English), PVR with test results (sensitivity, resolution, mass accuracy, retention time stability), certificate of refurbishment, and software license certificate.
Can the system be integrated into an existing LIMS environment?
Yes—GCMSsolution supports ASTM E1384-compliant ASCII export formats (.txt, .csv) and optional ODBC connectivity for direct LIMS data ingestion.
What warranty and service support are provided?
A 12-month comprehensive parts-and-labor warranty is included, backed by on-site technical support and remote assistance from certified Shimadzu-trained engineers across North America and EU regions.