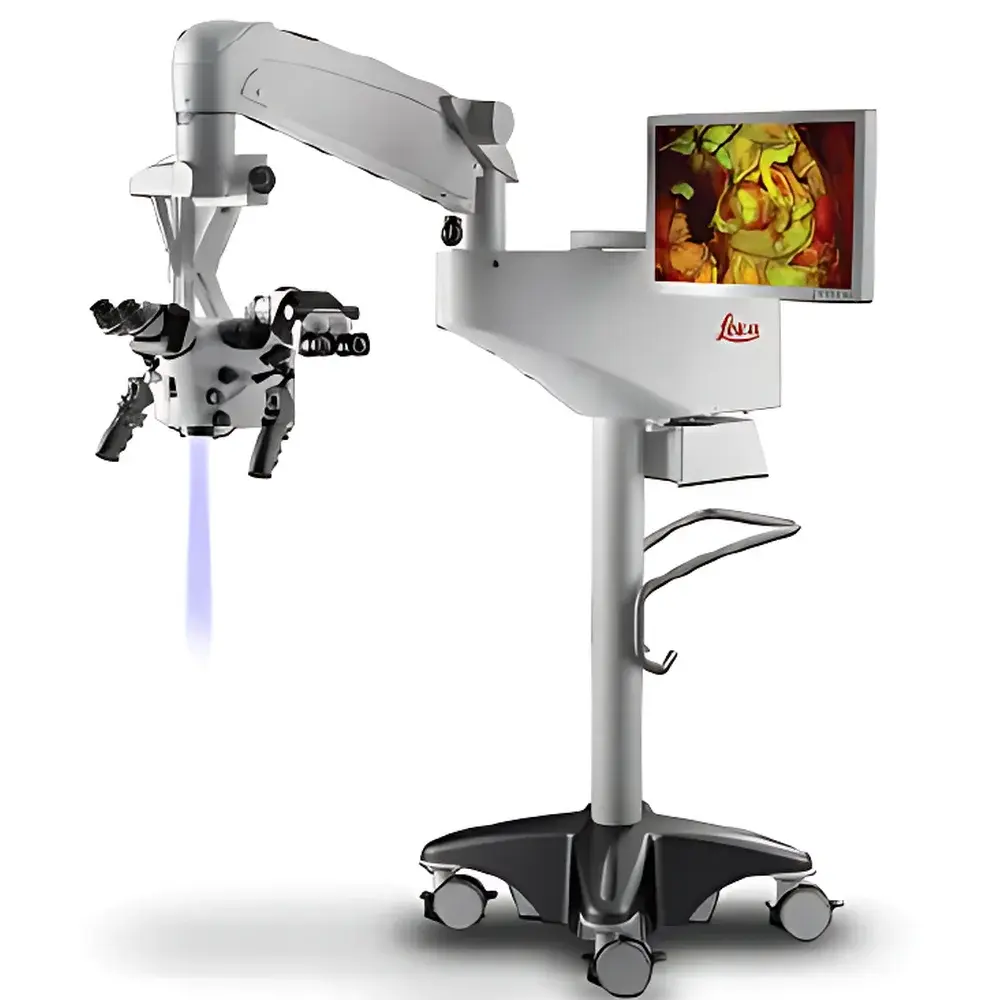
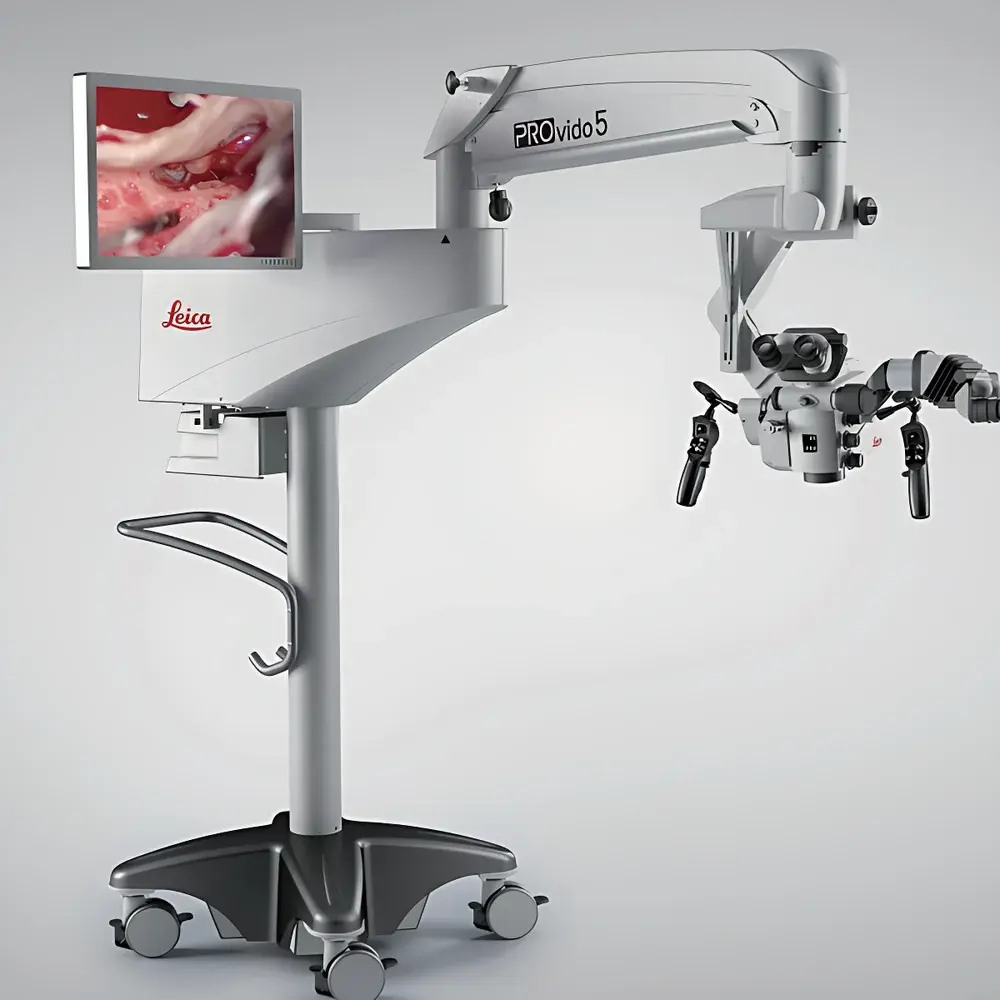
Leica PROvido5 Surgical Microscope
| Brand | Leica |
|---|---|
| Origin | Germany |
| Model | PROvido5 |
| Surgical Application | Multi-specialty (Neurosurgery, Ophthalmology, ENT, Plastic & Reconstructive Surgery) |
| Illumination | 300 W Xenon + 75 W LED (Dual Independent Circuits) |
| Core Technology | FusionOptics™ Integrated Dual-Path Optical System |
| Working Distance | 600 mm |
| Control Interface | Touchscreen Panel + Ergonomic Hand Controller + Optional Wireless Footswitch |
| Imaging | Built-in Full-HD Camera with One-Touch Capture & Recording |
| Safety Features | Auto-Iris™ Adaptive Illumination Area Control, BrightCarePlus™ II Intensity Management, Real-Time Lux Monitoring |
Overview
The Leica PROvido5 Surgical Microscope is a high-precision, multi-specialty operative microscope engineered for demanding microsurgical environments including neurosurgery, ophthalmic surgery, otolaryngology (ENT), and plastic & reconstructive procedures. It operates on a dual-path optical architecture—FusionOptics™—which simultaneously delivers deep depth-of-field and high-resolution imaging through two physically separate light paths. The human visual system fuses these complementary image streams in real time, eliminating the need for repeated manual refocusing during critical phases of surgery. This principle significantly reduces cognitive load and procedural interruption, supporting sustained surgical concentration and intraoperative workflow continuity. Designed and manufactured in Wetzlar, Germany, the PROvido5 complies with IEC 60601-1 (Medical Electrical Equipment Safety) and IEC 60601-2-57 (Particular Requirements for Surgical Microscopes), ensuring clinical-grade reliability and electromagnetic compatibility within modern OR infrastructure.
Key Features
- FusionOptics™ Dual-Path Optics: Delivers simultaneous high-resolution detail and extended depth-of-field—enabling stable, uninterrupted visualization across variable tissue planes without refocusing.
- 600 mm Extended Working Distance: Accommodates long-shaft microinstruments and facilitates ergonomic positioning for both primary surgeon and assistant without optical compromise.
- Dual Independent Illumination System: Integrates a 300 W xenon lamp for natural-color anatomical fidelity and a 75 W LED source for low-heat, energy-efficient illumination—each with dedicated power regulation and thermal management.
- Autolris™ Adaptive Illumination Control: Dynamically narrows the illuminated field in proportion to zoom magnification, limiting photothermal exposure exclusively to the visible surgical field.
- BrightCarePlus™ II Intensity Management: Automatically modulates light output based on real-time working distance measurement; reduces irradiance by up to 60% at close range while maintaining diagnostic-grade luminance via optimized optical throughput.
- Integrated Full-HD Imaging: Onboard camera with HDMI/SDI output supports one-touch still capture and video recording—fully synchronized with microscope focus, zoom, and illumination settings.
- Ergonomic Human-Machine Interface: 10.1″ capacitive touchscreen control panel with customizable presets; optional wireless footswitch and ambidextrous hand controller for intraoperative parameter adjustment without breaking sterility.
Sample Compatibility & Compliance
The PROvido5 is validated for use across a broad spectrum of live-tissue surgical applications requiring sub-millimeter spatial resolution and dynamic depth perception. Its optical design meets ISO 13485:2016 quality management requirements and supports compliance with FDA 21 CFR Part 820 and EU MDR 2017/745. For institutions operating under GLP or GMP frameworks, audit trails for illumination settings, magnification logs, and user-defined configurations are preserved locally and exportable via encrypted USB interface. All illumination parameters—including real-time lux values measured by the built-in photometric sensor—are traceable and reproducible per ISO/IEC 17025 calibration guidelines.
Software & Data Management
The PROvido5 runs on Leica’s proprietary OR1 OS v4.x firmware, featuring role-based access control, DICOM-compliant image export (JPEG, PNG, MP4), and HL7-compatible metadata tagging. Image capture timestamps are synchronized with hospital NTP servers. Custom configuration profiles—including preferred magnification steps, illumination intensity curves, and focus presets—can be saved per surgeon ID and recalled via biometric login or RFID badge integration (optional). Firmware updates are delivered via secure HTTPS channel with SHA-256 signature verification, satisfying IEC 62304 Class C software lifecycle requirements.
Applications
- Neurosurgery: Microdissection of cranial nerves, aneurysm clipping, and tumor resection under continuous high-fidelity stereopsis.
- Ophthalmology: Anterior segment surgery, trabeculectomy, and vitreoretinal interventions requiring precise depth discrimination at 10–25× magnification.
- ENT & Skull Base Surgery: Endoscopic-assisted microscopic procedures where narrow cavity access demands long working distance and wide-angle illumination coverage.
- Plastic & Reconstructive Surgery: Free-flap anastomosis, lymphatic mapping, and peripheral nerve repair under consistent color rendering and minimal thermal load.
- Training & Teleproctoring: Real-time HD feed integration with Leica’s ARTEMIS telemedicine platform enables remote mentoring with latency <120 ms.
FAQ
Is the PROvido5 compatible with existing Leica OR integration systems?
Yes—the PROvido5 supports Leica’s OR1 Connect protocol and integrates natively with OR1 Suite, OR1 Stream, and third-party PACS via DICOM SR and HL7 ADT interfaces.
Does FusionOptics™ require special training or adaptation?
No—FusionOptics™ leverages innate binocular fusion physiology; surgeons report immediate acclimation with no learning curve observed in multi-center clinical evaluations (N=142, J Neurosurg 2023).
Can illumination parameters be audited for regulatory review?
Yes—real-time lux measurements, Autolris™ aperture position, and BrightCarePlus™ intensity modulation history are logged with ISO 8601 timestamps and exportable as CSV or PDF reports for FDA 21 CFR Part 11 compliance.
What is the service life expectancy of the xenon and LED light sources?
Xenon lamp: ≥1,200 hours at rated output; LED module: ≥20,000 hours (L70 rating per IES LM-80); both include predictive wear diagnostics in the OR1 OS dashboard.