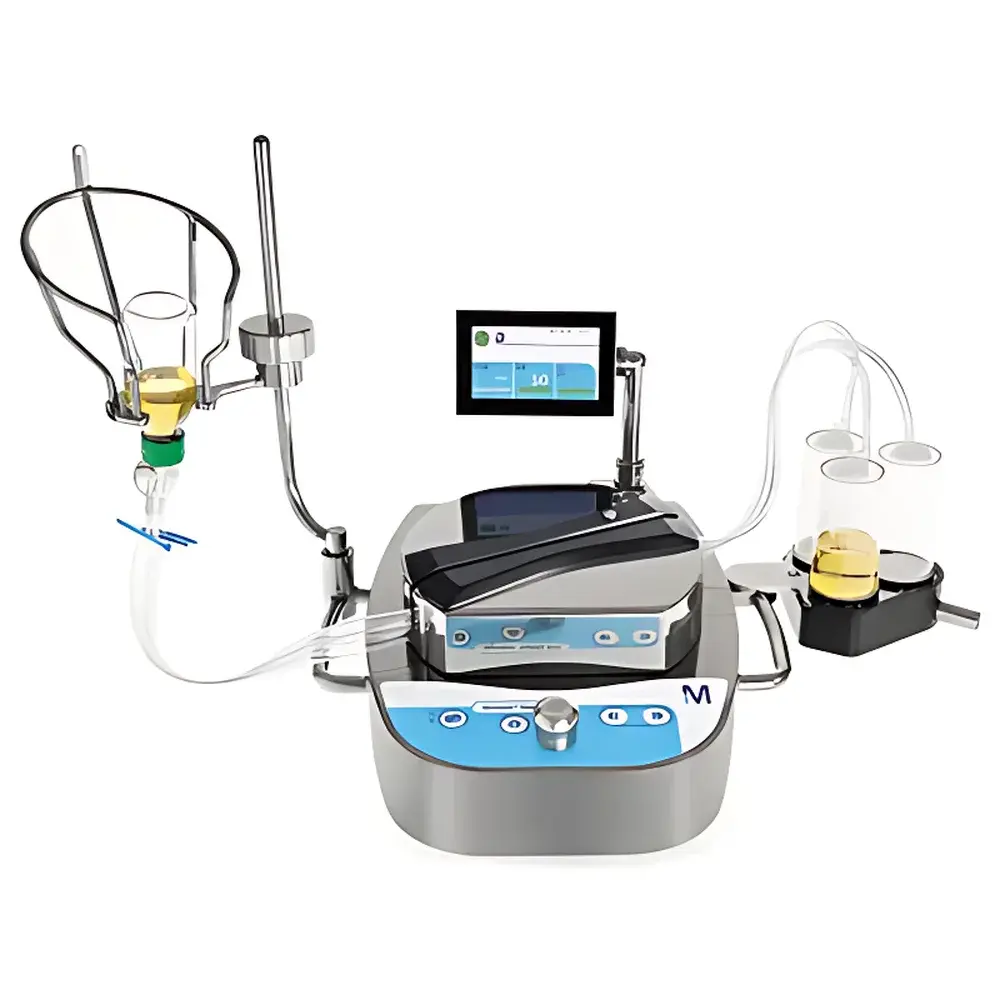
Merck Millipore Steritest™ Symbio Intelligent Sterility Testing System with 3-Canister Configuration
| Brand | Millipore |
|---|---|
| Origin | France |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Steritest™ Symbio with 3-canisters |
| Instrument Type | Fully Automated Sterility Testing System |
| Compliance | Designed for ISO 13408-1, USP <71>, EP 2.6.1, and FDA Aseptic Processing Guidance compliance |
| Software | Embedded touchscreen interface with audit trail, method versioning, and user access control (21 CFR Part 11 ready) |
| Filtration Architecture | Positive-pressure, dual-mode (constant pressure / adaptive flow rate reduction) per canister |
| Display | 11 cm (4.3 in) color LCD with tilt adjustment, multi-language support (EN/FR/DE/IT/ES/PT/JP/CN) |
| Method Storage | Up to 250 programmable sterility test protocols with revision tracking |
| Ergonomics | Low-profile design (≤28 cm height), glove-compatible buttons, adjustable bottle holder, passive cooling, particle-free enclosure |
Overview
The Merck Millipore Steritest™ Symbio Intelligent Sterility Testing System is an engineered solution for regulatory-compliant, fully automated membrane filtration-based sterility testing in pharmaceutical, biotechnology, and medical device manufacturing environments. Built upon the foundational principles of controlled positive-pressure filtration and gravimetric flow regulation, the system eliminates manual intervention during critical filtration, rinsing, and sample transfer steps—thereby minimizing operator-induced variability and contamination risk. Unlike legacy vacuum-driven or manually timed systems, Steritest™ Symbio employs a microprocessor-controlled peristaltic pump architecture with real-time pressure monitoring and adaptive speed modulation. Each of its three independent canister channels operates with synchronized timing, precise volume delivery (±1.5% accuracy across 10–500 mL ranges), and automatic transition between filtration, rinse, and elution phases. The system is purpose-built for integration into Grade A environments—including laminar airflow hoods, isolators, and restricted access barrier systems (RABS)—and conforms to the mechanical and operational requirements outlined in ISO 13408-1, USP , European Pharmacopoeia 2.6.1, and FDA’s Guidance for Industry on Sterile Drug Products Produced by Aseptic Processing.
Key Features
- Triple-canister configuration enabling parallel processing of up to three samples or sequential multi-step protocols (e.g., product + negative control + positive control) without cross-contamination risk.
- Dual-pressure operation mode: constant-pressure mode ensures uniform filter loading; adaptive-flow mode automatically reduces pump speed upon detection of increased resistance—preserving microbial viability and preventing filter clogging.
- Low-profile mechanical design (height ≤28 cm) optimized for seamless placement under laminar airflow workstations and within isolator glove ports—maximizing usable workspace and minimizing turbulence disruption.
- Passively cooled, hermetically sealed housing constructed from electro-polished stainless steel and USP Class VI-certified polymers—validated to emit <0.1 particles ≥0.5 µm/m³ under ISO 14644-1 Class 5 conditions.
- Glove-compatible human-machine interface featuring tactile feedback buttons, tilt-adjustable 4.3-inch color LCD, and eight-language support (English, French, German, Italian, Spanish, Portuguese, Japanese, Simplified Chinese).
- Embedded method management engine supporting up to 250 stored protocols, each with revision-controlled metadata (e.g., author, date, approval status), automatic parameter recall (flow rate, rinse volume, dwell time), and integrated validation of Steritest™ single-use filter cassette compatibility.
Sample Compatibility & Compliance
Steritest™ Symbio accommodates standard 47 mm and 50 mm diameter sterile filtration cassettes—including Merck’s Steritest™ Symbio-specific cassettes with pre-sterilized 0.45 µm or 0.22 µm PVDF membranes—and supports aqueous, oily, low-surface-tension, and moderately viscous pharmaceutical formulations (up to 25 cP). All wetted components comply with USP and cytotoxicity and systemic injection testing requirements. The system meets ISO 13408-1 Annex B criteria for equipment qualification, and its firmware architecture supports IQ/OQ documentation packages aligned with ASTM E2500-13 and EU GMP Annex 15. Audit trail functionality records all user actions, parameter changes, and method executions with immutable timestamps—fully compliant with 21 CFR Part 11 requirements when deployed with appropriate network authentication and electronic signature controls.
Software & Data Management
The embedded Steritest™ Symbio Control Software provides full lifecycle method governance: creation, validation, versioning, and execution—all accessible via the local touchscreen interface. Each protocol stores calibrated pump parameters, defined rinse sequences, and configurable alarm thresholds (e.g., pressure deviation >15%, duration exceedance >2%). Raw event logs—including start/stop times, actual vs. setpoint flow rates, and canister-specific pressure curves—are exportable in CSV format for external LIMS or QMS integration. Software updates are delivered via encrypted USB key with SHA-256 hash verification; no internet connectivity is required during operation. Revision history is retained locally for ≥10 years, satisfying GLP/GMP record retention mandates.
Applications
Steritest™ Symbio is routinely deployed in final container sterility release testing of injectables, ophthalmic solutions, and lyophilized products; environmental monitoring of cleanroom air and surface rinsates; sterility validation of sterilization processes (e.g., steam, gamma, ethylene oxide); and microbiological quality control of raw materials and excipients. Its precision flow control makes it especially suitable for low-bioburden products where recovery efficiency must be maximized—such as monoclonal antibody formulations and cell therapy intermediates—without compromising membrane integrity or microbial retention.
FAQ
Does Steritest™ Symbio require external compressed air or vacuum sources?
No. It operates exclusively via integrated brushless DC peristaltic pumps—eliminating dependency on facility utilities and associated validation burdens.
Can the system be qualified for use in regulated GMP environments?
Yes. Merck provides comprehensive IQ/OQ documentation templates, installation checklists, and performance verification protocols aligned with ASTM E2500-13 and EU Annex 15.
Is remote monitoring or data export supported?
Raw operational logs and method execution reports are exportable via USB 2.0 port in timestamped CSV format; no proprietary software or cloud connection is required.
What maintenance is required beyond routine cleaning?
Annual calibration of pressure sensors and flow verification using NIST-traceable standards is recommended; pump tubing replacement intervals are tracked automatically and displayed on-screen.
How does the system ensure method reproducibility across operators?
All critical parameters—including flow rate, rinse volume, hold time, and pressure thresholds—are locked within the selected protocol; deviations trigger audible/visual alarms and halt execution until acknowledged by authorized personnel.