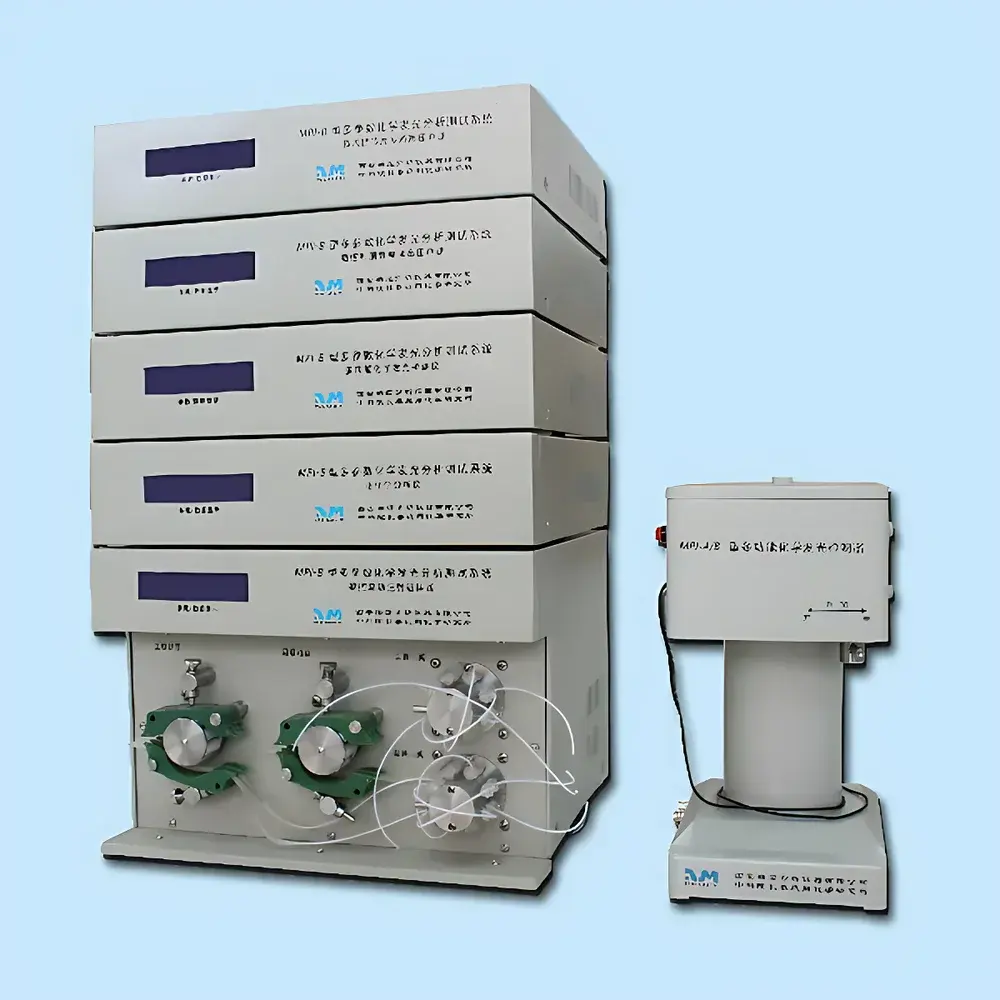
DMT 620M Wire Myograph System
| Origin | Denmark |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | 620M |
| Pricing | Upon Request |
Overview
The DMT 620M Wire Myograph System is a fourth-generation, high-precision ex vivo vascular tension measurement platform engineered for quantitative pharmacological and physiological assessment of isolated blood vessels, airways, and gastrointestinal smooth muscle rings or strips. Based on the well-established isometric wire myography principle, the system measures contractile and relaxant responses under controlled mechanical preload and physiological bath conditions. As the direct successor to the DMT 610M series, the 620M integrates enhanced thermal stability, modular bath architecture, and expanded sample handling flexibility—making it the most widely adopted platform in academic cardiovascular research labs, contract research organizations (CROs), and pharmaceutical preclinical departments across North America and Europe.
Key Features
- Four independent, parallel measurement channels: Enables simultaneous real-time recording of up to four tissue samples—significantly improving throughput for dose–response profiling, comparative pharmacology, and inter-laboratory reproducibility studies.
- Dual-mounting hardware configuration: Includes precision-machined clamp-style holders for microvessels (60–450 µm diameter) and needle-based holders compatible with larger conduit vessels (up to 10 mm outer diameter), ensuring optimal mechanical coupling and minimal tissue distortion.
- Integrated thermostatic control: Each stainless-steel bath features an embedded heating element, external temperature probe feedback (±0.1 °C accuracy), and independent gas inlet valves—supporting stable physiological temperatures from ambient up to 45 °C under continuous superfusion.
- Configurable force transduction range: User-selectable full-scale sensitivity options (±200 / ±400 / ±800 / ±1600 mN) with 0.1 mN resolution, calibrated via semi-automatic weight verification routine compliant with ISO/IEC 17025 traceability requirements.
- Automated fluid management support: Optional DMT 625FS buffer auto-filler enables programmable, synchronized perfusion of all four baths—reducing manual intervention and minimizing inter-bath variability during long-duration experiments (e.g., 12-hour viability assays).
Sample Compatibility & Compliance
The DMT 620M accommodates a broad spectrum of native and engineered tissues: small resistance arteries (mesenteric, cerebral, coronary), large elastic arteries (aorta, carotid), bronchial segments, ileal or colonic rings, and skeletal muscle strips. Tissue viability is maintained for ≥12 hours post-isolation when operated within validated physiological parameters (37 °C, 95% O2/5% CO2, Krebs-Henseleit buffer). The system conforms to international standards for ex vivo functional testing—including ASTM F2961 (Standard Guide for In Vitro Pharmacological Testing of Vascular Tissues), ISO 10993-5 (biocompatibility of stainless-steel bath components), and supports GLP-compliant data acquisition workflows per FDA 21 CFR Part 11 when paired with validated software modules.
Software & Data Management
Data acquisition is performed via USB 2.0 interface using DMT’s proprietary MyoVIEW™ v5.x software suite—designed for regulatory-grade experimental design, real-time visualization, and audit-trail-enabled analysis. The software provides synchronized multi-channel analog output (2.5 V full scale, ±10 V range), timestamped digital event markers (e.g., drug additions, electrical pulses), and automated calculation of EC50/IC50, Emax, pD2, and Hill coefficients. Raw data exports are compliant with HDF5 and CSV formats; metadata fields include operator ID, calibration logs, bath temperature history, and user-defined protocol annotations—facilitating seamless integration into LIMS and electronic lab notebook (ELN) environments.
Applications
- Characterization of vasoactive compound efficacy and mechanism of action (e.g., endothelin receptor antagonists, Rho-kinase inhibitors)
- Assessment of endothelial function and NO bioavailability in hypertension, diabetes, and atherosclerosis models
- Functional phenotyping of genetically modified murine vasculature (e.g., KO/KI strains)
- Preclinical safety evaluation of novel biologics and nanotherapeutics on vascular reactivity
- Standardized contractility assays for ICH S7B and FDA guidance on cardiovascular risk assessment
FAQ
What is the minimum vessel diameter supported by the DMT 620M?
The system supports microvessels down to 60 µm outer diameter using the clamp-style holder.
Can the 620M be used for non-vascular tissues such as trachea or intestine?
Yes—its modular mounting system and adjustable preload protocols are validated for airway smooth muscle and GI tract preparations.
Is the system compatible with electrical field stimulation (EFS)?
Yes, optional CS200 pulse/current stimulators and EFS-compatible plastic clamp holders are available for neurogenic response studies.
Does the DMT 620M meet regulatory requirements for GLP-compliant studies?
When configured with MyoVIEW™ v5.x in audit mode and paired with documented calibration and maintenance records, it satisfies core GLP data integrity criteria per OECD Principles of Good Laboratory Practice.
How is bath temperature stability ensured across all four channels?
Each bath has an independent PID-controlled heater, external Pt100 temperature sensor, and user-definable setpoint ramping—achieving ±0.1 °C uniformity over 12-hour experiments.