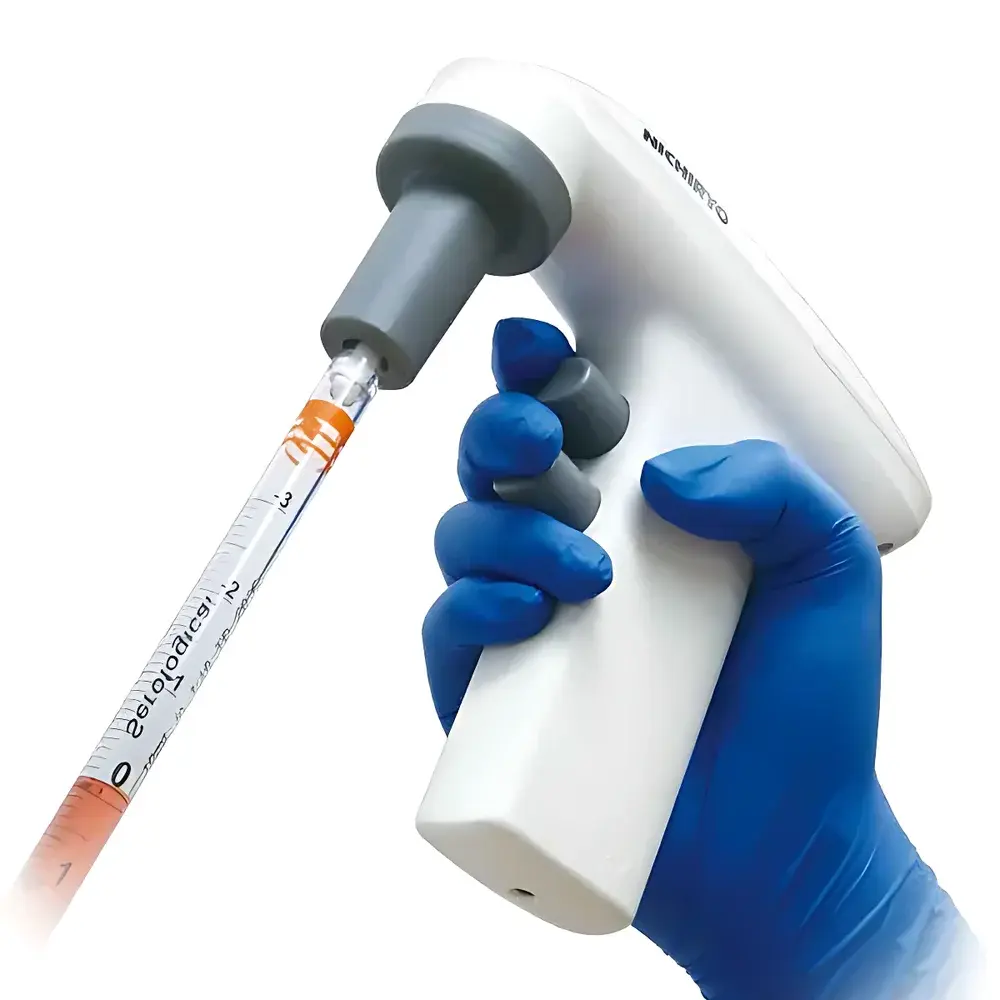
Nichiryo Pipette Mate NEO Electric Single-Channel Pipette Controller
| Brand | Nichiryo |
|---|---|
| Origin | Japan |
| Model | Pipette Mate NEO |
| Instrument Type | Single-Channel Electric Pipette Controller |
| Power Source | Rechargeable Lithium-Ion Battery (3.6 V, 600 mAh) |
| Operating Time | ~1700 dispenses per full charge (4-hour charging time) |
| Pipette Compatibility | 1–100 mL plastic or glass serological pipettes |
| Filter | Hydrophobic 0.2 µm (20 mm diameter) |
| Compliance | CE, RoHS |
| Autoclavable Components | Nosecone, Silicone Holder, Filter |
| Weight | 167 g |
| Ergonomic Design | Self-standing, UV-resistant housing |
| Speed Control | Dial-type adjustable suction/dispense speed |
Overview
The Nichiryo Pipette Mate NEO is an ergonomically engineered electric single-channel pipette controller designed for high-frequency, repetitive liquid handling tasks in ISO-certified cleanrooms, GLP-compliant laboratories, and clinical QC environments. Unlike manual pipetting aids or basic battery-powered controllers, the Pipette Mate NEO employs a precision brushless DC motor coupled with a digitally stabilized torque control system to deliver consistent aspiration and dispensing across its full operational range (1–100 mL). Its core architecture follows the positive-displacement principle—where motor-driven piston movement generates controlled negative pressure within the pipette barrel—ensuring minimal operator-induced variability and eliminating reliance on user thumb force or wrist fatigue. The device operates without external air compressors or vacuum manifolds, making it ideal for benchtop deployment in biosafety cabinets, laminar flow hoods, and mobile lab setups where portability and electrical independence are critical.
Key Features
- Ultra-Lightweight & Self-Standing Design: Weighing only 167 g, the Pipette Mate NEO features a low-center-of-gravity base geometry that enables stable upright positioning without requiring a dedicated holder—reducing bench clutter and minimizing cross-contamination risk during multi-step workflows.
- Long-Duration Lithium-Ion Power System: Integrated 3.6 V / 600 mAh rechargeable battery supports approximately 1700 complete pipetting cycles per full 4-hour charge. Battery status is indicated via dual LED indicators (charging/operation), and the battery cover (Cat. No. 00-PM-N003) is tool-free replaceable for field maintenance.
- Dial-Type Variable Speed Control: A tactile rotary dial allows continuous, analog adjustment of both aspiration and dispensing speeds—from gentle, dropwise delivery for viscous or volatile samples to rapid bulk transfer—without mode switching or software navigation.
- UV-Resistant Housing & Cleanroom Compatibility: Constructed from medical-grade ABS polymer with UV-stabilized additives, the unit maintains structural integrity and surface integrity under prolonged exposure to germicidal UV-C (254 nm) irradiation commonly used in biosafety cabinet decontamination protocols.
- Fully Autoclavable Critical Components: Nosecone (Cat. No. 00-PM-N005), silicone pipette holder (Cat. No. 00-PM-N013), and hydrophobic 0.2 µm filter (Cat. No. 00-PM-N100) are rated for repeated autoclaving at 121 °C, 15 psi, 20 min—ensuring compliance with ISO 13485 and AAMI ST79 sterility assurance requirements.
Sample Compatibility & Compliance
The Pipette Mate NEO accommodates standard conical-tip serological pipettes—both disposable plastic and reusable borosilicate glass—within the 1 mL to 100 mL volume range. It does not require proprietary pipettes; compatibility is verified per ISO 8655-3 (volumetric accuracy testing for pipette controllers) and ASTM E2792 (standard guide for selection and use of pipette controllers). All electrical components meet CE marking requirements under the Low Voltage Directive (2014/35/EU) and Electromagnetic Compatibility Directive (2014/30/EU). RoHS 3 (EU Directive 2015/863) compliance ensures restricted substance thresholds (e.g., Pb, Cd, Hg, Cr⁶⁺, PBB, PBDE) are fully satisfied. While not FDA 510(k)-cleared as a medical device, its design aligns with IEC 61010-1 safety standards for laboratory electrical equipment.
Software & Data Management
The Pipette Mate NEO is a standalone hardware controller with no embedded firmware, cloud connectivity, or digital logging capability—intentionally designed to eliminate data integrity concerns associated with unvalidated software layers. This architecture supports audit-ready operation in regulated environments governed by 21 CFR Part 11 (FDA), EU Annex 11, and ISO/IEC 17025:2017 Clause 7.7 (traceability of measurement equipment). All calibration and performance verification records—including gravimetric testing per ISO 8655-6—are maintained externally using paper-based or LIMS-integrated SOPs. Optional travel chargers (US: 00-PM-N024; UK: 00-PM-N025 + base unit 00-PM-N030; AU: 00-PM-N026; EU: 00-PM-N027) support universal input (100–240 V, 50/60 Hz), enabling global deployment without voltage conversion hardware.
Applications
- Cell culture media preparation and passaging in Class II BSCs
- Serial dilution workflows in ELISA, PCR setup, and microbiological plating
- Reagent dispensing for hematology analyzers and automated immunoassay platforms
- Quality control sampling in pharmaceutical water systems (PW, WFI)
- Environmental water analysis involving turbid or particulate-laden samples where filter integrity is critical
- Teaching laboratories requiring durable, low-maintenance pipetting aids compliant with undergraduate safety standards
FAQ
Is the Pipette Mate NEO compatible with Gilson, Eppendorf, or BrandTech pipettes?
Yes—it accepts any standard 1–100 mL conical-tip serological pipette meeting ISO 8655-1 dimensional tolerances, regardless of manufacturer.
Can the 0.2 µm hydrophobic filter be replaced with a 0.45 µm alternative?
No—the device is validated exclusively with the supplied 20 mm × 0.2 µm hydrophobic PTFE filter (Cat. No. 00-PM-N100); substitution may compromise aerosol containment and void warranty.
Does the unit require periodic factory recalibration?
No—calibration is user-performed gravimetrically per ISO 8655-6; no firmware or sensor drift compensation is implemented, ensuring long-term metrological stability.
What is the recommended cleaning protocol between uses with infectious samples?
Wipe exterior surfaces with 70% ethanol; autoclave nosecone, silicone holder, and filter at 121 °C for 20 minutes; do not immerse main body in liquid or subject to steam sterilization.
Is the battery replaceable by the end user?
Yes—battery replacement requires only removal of the snap-fit cover (Cat. No. 00-PM-N003); replacement batteries are available as service parts under Nichiryo’s global spare parts program.