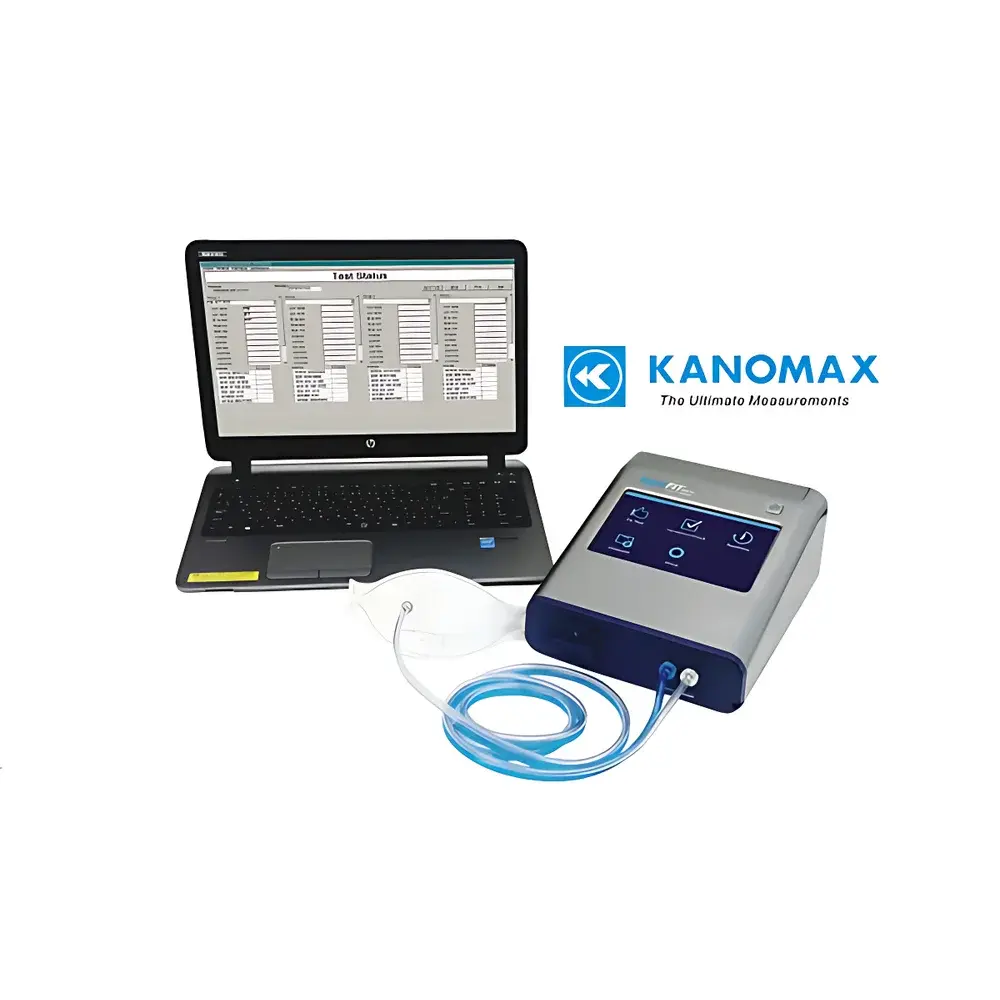
Kanomax AccuFIT 9000 Respirator Fit Testing System
| Brand | Kanomax |
|---|---|
| Origin | Japan |
| Model | 3000-0C |
| Compliance | GB 2626–2019, GB 19083–2010, OSHA 29 CFR 1910.134, CSA Z94.4, EN 13274-1/EN 13274-7 |
Overview
The Kanomax AccuFIT 9000 Respirator Fit Testing System (Model 3000-0C) is a precision-engineered, quantitative fit testing instrument designed to measure the real-world performance of respirators—including N95, KN95, KP100, P3, HEPA, and reusable half/full-facepiece respirators—by calculating the Total Inward Leakage (TIL) ratio under controlled challenge conditions. Utilizing condensation nuclei counter (CNC) technology, the system quantifies aerosol particle concentration both outside and inside the respirator facepiece during standardized test exercises (e.g., normal breathing, deep breathing, head side-to-side, head up-and-down, grimace, and talking), enabling objective, repeatable assessment of facial seal integrity. Unlike qualitative methods reliant on subjective sensory response, the AccuFIT 9000 delivers numerical fit factors (FF) traceable to NIST-calibrated reference aerosols, satisfying regulatory requirements for quantitative fit testing (QNFT) under OSHA 29 CFR 1910.134, CSA Z94.4, and China’s GB 2626–2019 and GB 19083–2010 standards. Its CNC-based optical detection principle ensures high sensitivity across sub-micron particle sizes (down to 20 nm), with minimal inter-unit variability and robust immunity to environmental humidity fluctuations.
Key Features
- Quantitative fit testing platform compliant with ISO 16900-1:2016, ASTM F1885–22, and EN 13274-1/7 for respirator leakage evaluation
- Condensation Nucleus Counter (CNC) detection technology with dual-channel real-time aerosol concentration measurement (ambient vs. intramask)
- Automated calculation of Fit Factor (FF) and Total Inward Leakage (TIL) per test subject, supporting pass/fail determination per regulatory thresholds (e.g., FF ≥ 100 for N95/KN95; FF ≥ 500 for full-facepieces)
- Pre-programmed test protocols aligned with OSHA, CSA, and GB standards—including 7-exercise sequence with configurable timing and motion profiles
- Multi-language interface (English, Japanese, Chinese, Korean, German) with intuitive touchscreen operation and on-device data review
- Flexible connectivity: USB 2.0, 10/100 Mbps Ethernet, and optional Wi-Fi module—enabling centralized control of up to four units from a single PC via dedicated software
- Modular design supporting both standalone operation and networked deployment in occupational health clinics, hospital infection control units, or industrial hygiene labs
Sample Compatibility & Compliance
The AccuFIT 9000 accommodates a broad spectrum of respiratory protection devices: disposable filtering facepiece respirators (FFRs) including N95, KN90, KN95, KP100, FFP2, FFP3; reusable elastomeric half-mask and full-facepiece respirators; and powered air-purifying respirators (PAPRs) with tight-fitting hoods or facepieces. All testing procedures adhere to internationally recognized performance criteria. For example, under GB 2626–2019, the instrument verifies that the overall TIL for a cohort of 10 subjects does not exceed 8% for KN95/KP95-class respirators, or 2% for KN100/KP100. Similarly, per OSHA 29 CFR 1910.134, it validates minimum fit factors of 100 for half-masks and 500 for full-facepieces. The system supports audit-ready documentation required for GLP/GMP-aligned occupational safety programs and FDA-regulated healthcare environments (e.g., surgical mask validation under USP particulate matter guidance).
Software & Data Management
The AccuFIT 9000 integrates with Kanomax’s proprietary FitTest Manager software (Windows-based), which provides comprehensive data acquisition, analysis, and reporting capabilities. Each test session generates a timestamped, user-authenticated record containing subject ID, respirator model, test date/time, ambient conditions (temperature, RH), individual exercise results, calculated FF/TIL values, and pass/fail status. All data fields support export to CSV or PDF formats for integration into enterprise EHS platforms. The software implements role-based access control, electronic signature functionality, and full 21 CFR Part 11-compliant audit trails—including immutable logs of test modifications, operator logins, and calibration events—to meet regulatory inspection requirements in pharmaceutical manufacturing, clinical research, and public health infrastructure.
Applications
- Hospital infection prevention programs validating surgical N95 and medical-grade KN95 respirators for frontline staff
- Occupational safety departments conducting mandatory annual respirator fit testing for workers exposed to silica, asbestos, metal fumes, or bioaerosols
- Fire department and emergency response teams certifying SCBA facepiece seal integrity before hazardous material operations
- Respirator manufacturers performing R&D validation and pre-market conformity testing against GB, EN, and ASTM specifications
- National metrology institutes and third-party certification labs executing accredited fit testing services per ISO/IEC 17025
- University and government research laboratories studying facial anthropometry–respirator interface dynamics and leakage pathway modeling
FAQ
What aerosol challenge agent does the AccuFIT 9000 use?
It employs ambient room air as the natural polydisperse aerosol source—no artificial challenge agents (e.g., sodium chloride, corn oil, or isoamyl acetate) are required, eliminating consumables and cross-contamination risk.
Can the system test respirators with exhalation valves?
Yes—its dual-sensor architecture independently measures inhalation- and exhalation-phase leakage, ensuring accurate TIL calculation regardless of valve configuration.
Is calibration traceable to national standards?
All CNC sensors are factory-calibrated using NIST-traceable polystyrene latex (PSL) nanoparticles and verified annually per ISO 17025 procedures.
Does it support remote monitoring during multi-station testing?
Yes—via Ethernet or Wi-Fi, operators can monitor live FF trends, pause/resume tests, and trigger manual overrides across up to four synchronized instruments from one workstation.
How is subject data protected under GDPR or HIPAA?
Data remains on-premise unless explicitly exported; no cloud storage or telemetry transmission occurs without explicit administrator consent and encrypted transfer protocols.