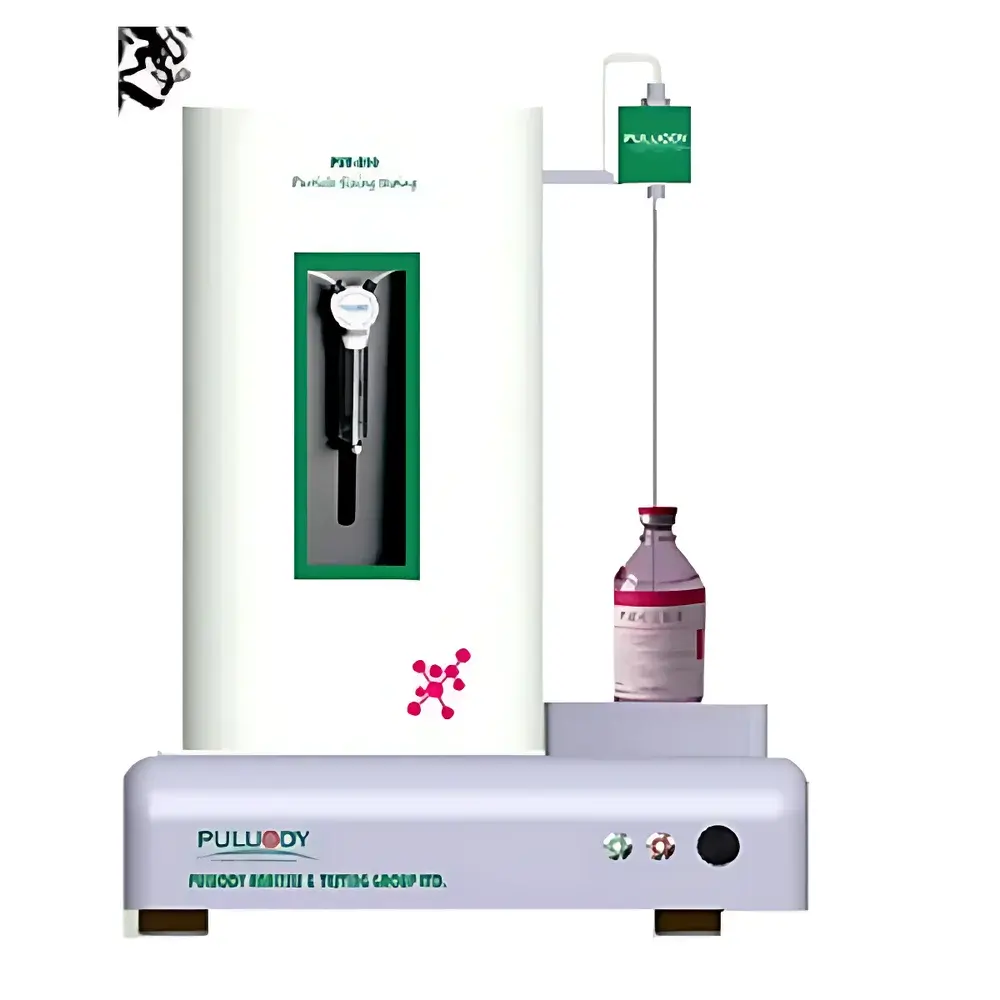
PULUODY PMF-2801.1 Online & Offline Ultra-Pure Water Particle Counter
| Brand | PULUODY |
|---|---|
| Origin | Shaanxi, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | PMF-2801.1 |
| Instrument Type | Optical Particle Counter |
| Detection Principle | Dual-Laser Narrow-Beam Light Scattering |
| Sensor Generation | 8th-Generation Dual-Laser Sensor |
| Measurement Range | 0.1–1.0 µm (standard), customizable 1–100 µm or 4–70 µm(c), down to 0.1 µm(c) |
| Sampling Accuracy | <±1% |
| Counting Accuracy | ±3% typical |
| Coincidence Limit | ≤2.5% at 1000 particles/mL |
| Flow Control | Precision Piston Pump + Electromagnetic Flow Regulation |
| Calibration Standards | ISO 21501-4, USP <788>, EP 2.9.19, ChP 2015/2020, GB 8368, JJG 1061 |
| Software | V8.3 Integrated Analysis & Calibration Software (PC & Standalone Touch Interface) |
| Output | 4–20 mA Analog Signal, Optional Alarm Relay, iPad-Compatible Data Acquisition |
| Compliance Mode | Separated Civil/Military Standard Configuration |
| Display | Color LCD Touchscreen + Optional Wireless Keyboard/Mouse |
| Power Input | 100–265 VAC, 50–60 Hz |
| Reporting | Particles per mL + ISO 4406 / NAS 1638 Contamination Code |
Overview
The PULUODY PMF-2801.1 Online & Offline Ultra-Pure Water Particle Counter is an optical particle counter engineered for high-sensitivity, trace-level particulate monitoring in ultra-pure water (UPW), semiconductor process chemicals, and critical liquid media. It operates on the principle of dual-laser narrow-beam light scattering—where two spatially separated laser sources illuminate the sample stream simultaneously, enabling improved signal-to-noise ratio, reduced coincidence error, and enhanced resolution for sub-micron particles. This 8th-generation sensor architecture is specifically optimized for low-conductivity, low-refractive-index liquids such as UPW (resistivity >18.2 MΩ·cm), hydrogen peroxide solutions, SC1/SC2 cleans, and photoresist solvents. The instrument supports both continuous online installation—integrated into recirculation loops or point-of-use lines—and offline portable operation via calibrated syringe or bottle sampling. Its design meets the stringent metrological requirements of microelectronics fabrication, pharmaceutical water systems, and advanced materials R&D laboratories where particle contamination at the 0.1 µm level directly impacts yield, device reliability, and regulatory compliance.
Key Features
- Dual-laser narrow-beam detection with independent wavelength alignment for simultaneous multi-size binning and improved sizing fidelity in transparent low-scatter media
- Precision piston pump combined with electromagnetic flow regulation ensures volumetric accuracy better than ±1% across 1–100 mL/min operational range
- Modular configuration supporting either fixed-mount online deployment or battery-assisted mobile offline use with integrated sample chamber and zero-contamination fluid path
- Real-time alarm output via 4–20 mA analog interface and programmable relay—configurable by size channel, cumulative count, or rate-of-change thresholds
- Separate calibration and analysis software environments (V8.3) prevent cross-interference between metrological verification and routine measurement workflows
- Compliance-ready firmware architecture with audit trail logging, user access levels, and electronic signature support aligned with FDA 21 CFR Part 11 expectations
Sample Compatibility & Compliance
The PMF-2801.1 is validated for use with ultra-pure water, deionized water, hydrogen peroxide (H₂O₂), ammonium hydroxide (NH₄OH), hydrochloric acid (HCl), sulfuric acid (H₂SO₄), and organic solvents including NMP and PGMEA—provided refractive index differences from water remain within ±0.05 RIU. It conforms to pharmacopeial standards including USP , EP 2.9.19, ChP 2015/2020, and JP 17, as well as industrial benchmarks ISO 21501-4 (calibration), ISO 4406 (contamination coding), and SEMI F63 (UPW particle monitoring). All factory calibrations are traceable to NIST-traceable polystyrene latex (PSL) reference standards, and optional on-site verification follows JJG 1061 national metrology verification procedures. The system supports GLP/GMP documentation packages, including IQ/OQ protocols and raw data export in CSV, PDF, and XML formats.
Software & Data Management
The V8.3 software suite provides dual-mode operation: a standalone embedded interface running on the instrument’s color touchscreen (with optional wireless keyboard/mouse), and a full-featured PC application for advanced reporting, trend analysis, and database integration. Data acquisition includes timestamped particle counts per size channel (e.g., ≥0.1 µm, ≥0.2 µm, ≥0.5 µm), calculated contamination codes (ISO 4406, NAS 1638), and statistical summaries (mean, SD, CV%). Raw pulse data is retained for retrospective reanalysis. Export functions comply with LIMS interoperability requirements; metadata fields include operator ID, sample ID, location tag, environmental conditions (temperature, pressure), and calibration certificate ID. Audit logs record all user actions—including parameter changes, calibration events, and report generation—with immutable timestamps and digital signatures.
Applications
This particle counter serves critical quality control functions across multiple high-tech sectors: real-time UPW loop monitoring in 300mm wafer fabs; post-filtration validation of 2 nm-rated membrane filters; cleanliness verification of silicon wafers and glass substrates after wet cleaning; particulate release testing of medical-grade tubing and IV bags per ISO 8536-4; and stability assessment of nanosuspensions and colloidal drug carriers. In R&D environments, it enables kinetic studies of particle agglomeration, filter breakthrough profiling, and leachables evaluation in single-use bioprocessing systems. Its adaptability to both conductive and non-conductive liquids—without requiring conductivity adjustment or dilution—makes it suitable for next-generation semiconductor chemistries and aqueous nanomaterial synthesis processes.
FAQ
What particle size ranges can the PMF-2801.1 detect?
It offers standard detection from 0.1 µm to 1.0 µm (with 0.1 µm(c) sensitivity), and supports custom size channels from 1 µm to 100 µm or 4 µm to 70 µm(c) upon configuration.
Is the instrument compliant with pharmaceutical water system validation requirements?
Yes—it satisfies USP , EP 2.9.19, and ChP 2020 particulate testing criteria, and supports IQ/OQ documentation, calibration traceability, and 21 CFR Part 11–compliant audit trails.
Can it operate continuously in-line without manual intervention?
Yes—its robust flow control system, self-diagnostic firmware, and analog alarm outputs enable unattended 24/7 online monitoring in UPW distribution networks and chemical delivery systems.
How is calibration verified during routine use?
Calibration is performed using NIST-traceable PSL standards per ISO 21501-4; field verification may be conducted using certified reference suspensions or through interlaboratory comparison protocols.
Does the system support integration with plant-wide SCADA or MES platforms?
Yes—via Modbus TCP/IP, OPC UA, or 4–20 mA analog output, with configurable data polling intervals and alarm state mapping to enterprise-level automation systems.