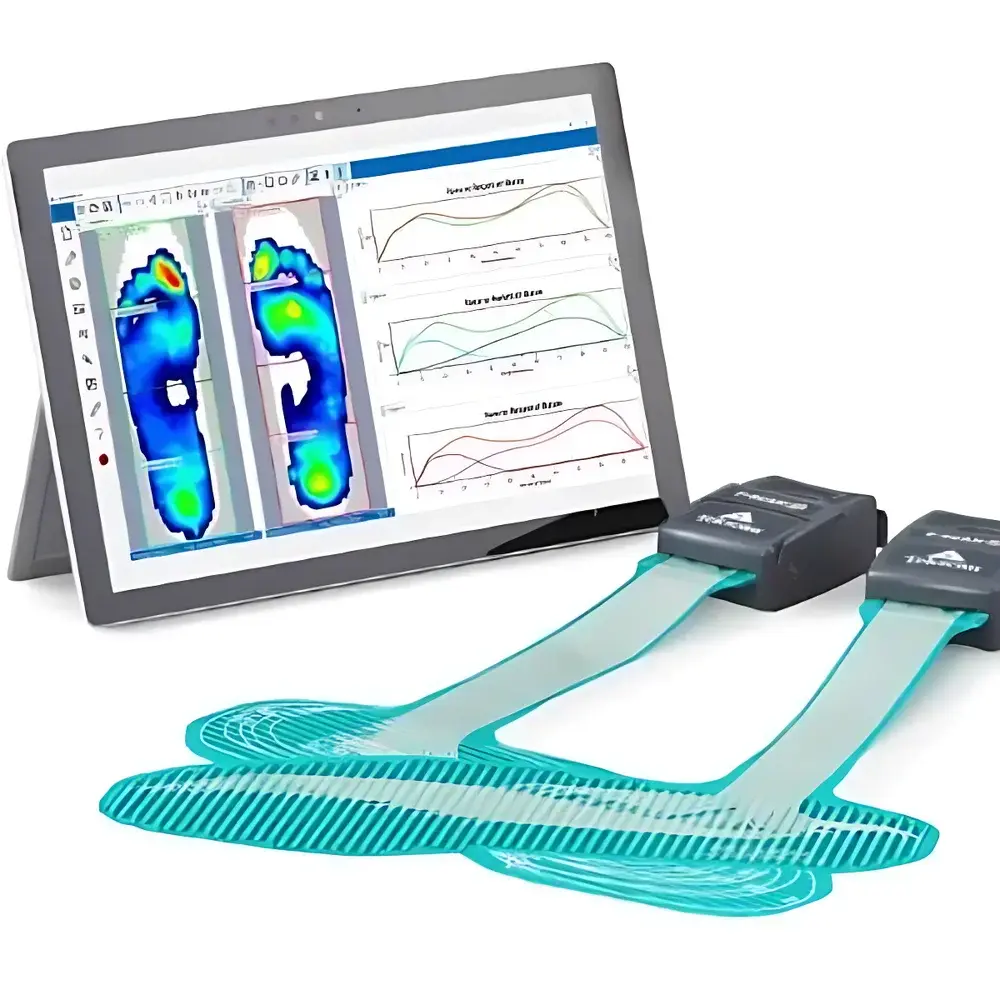
Tekscan F-Scan GO In-Shoe Plantar Pressure & Gait Analysis System
| Brand | Tekscan |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | F-Scan GO |
| Pricing | Available Upon Request |
Overview
The Tekscan F-Scan GO In-Shoe Plantar Pressure & Gait Analysis System is a wireless, high-fidelity physiological signal acquisition platform engineered for quantitative biomechanical assessment of dynamic foot function during unrestricted ambulation. Built upon capacitive thin-film sensor technology, the system measures plantar pressure distribution in real time using a calibrated array of micro-scale sensing elements embedded within an ultra-thin, conformable insole. Unlike tethered systems that constrain kinematic freedom or introduce motion artifacts, the F-Scan GO employs a fully integrated wireless architecture—eliminating cables, external data loggers, and proximity limitations—thereby preserving natural gait kinematics across diverse environments including clinical gait labs, rehabilitation clinics, sports training facilities, and field-based research settings. Its core measurement principle relies on capacitance variation induced by localized mechanical deformation under load, enabling sub-millisecond temporal resolution and spatially resolved pressure mapping at anatomically relevant foot regions (e.g., medial/lateral forefoot, midfoot, heel, hallux).
Key Features
- Fully wireless electronics with integrated Wi-Fi 802.11n connectivity for real-time streaming at up to 500 Hz sampling rate
- Onboard SD card storage (expandable) for autonomous long-duration recording without host dependency
- Ultra-lightweight (< 45 g), low-profile sensor insole (≤ 0.3 mm thickness) designed for minimal footwear interference and multi-session durability
- High-density capacitive sensor array delivering industry-leading spatial resolution (up to 16 sensors/cm²) and dynamic range (0–1000 kPa)
- Real-time synchronization capability via TTL-compatible external trigger input for concurrent integration with EMG, motion capture (Vicon, Qualisys), force plates, or respiratory monitoring systems
- IP65-rated electronics housing for resistance to dust and incidental moisture during active ambulation protocols
Sample Compatibility & Compliance
The F-Scan GO system accommodates standard athletic, orthopedic, and custom-molded footwear without modification. Sensor insoles are available in 12 anatomically scaled sizes (US Men’s 6–14, Women’s 5–12) and support both barefoot and socked conditions. All hardware components comply with FCC Part 15 Class B and CE RED directives for electromagnetic compatibility. Software workflows adhere to principles aligned with ISO 13485:2016 (medical device quality management) and support audit-trail-enabled data handling consistent with GLP and clinical research best practices. While not classified as a diagnostic medical device per FDA 21 CFR Part 892, the system meets performance criteria referenced in ASTM F3077-22 (Standard Guide for Biomechanical Testing of Footwear and Orthotics) and supports documentation required for IRB-approved human subject studies.
Software & Data Management
FootVIEW v5.x—the next-generation analysis software—provides a modular, touch-optimized interface compatible with Windows 10/11 and iOS tablets. It features dockable panels for synchronized waveform visualization (pressure-time, COP trajectory, force vector plots), region-of-interest (ROI) definition tools, and automated event detection (initial contact, toe-off). Raw binary data exports are supported in CSV, MATLAB (.mat), and C3D formats; metadata embedding includes session ID, calibration timestamp, sensor ID, and user-defined annotations. The software implements role-based access control, encrypted local database storage, and configurable export logs compliant with 21 CFR Part 11 requirements for electronic records and signatures when deployed in regulated environments.
Applications
- Clinical gait analysis for diabetic foot ulcer risk stratification and post-operative rehabilitation monitoring
- Evidence-based orthotic design validation and iterative intervention assessment (pre-/post-fitting pressure redistribution analysis)
- Sports biomechanics research—including sprint mechanics, jump landing asymmetry, and footwear performance benchmarking
- Geriatric mobility assessment targeting fall risk prediction through dynamic balance and weight-transfer metrics
- Prosthetic socket interface pressure mapping and alignment optimization
- Academic biomechanics education with portable lab modules for undergraduate and graduate curricula
FAQ
What is the maximum recommended continuous recording duration on internal SD storage?
With a 32 GB microSD card and 500 Hz sampling, typical continuous recording exceeds 45 minutes per session; duration scales linearly with card capacity and inversely with sampling rate.
Is sensor calibration traceable to NIST standards?
Yes—each sensor batch undergoes factory calibration against certified dead-weight test loads; calibration certificates include uncertainty budgets and are provided with shipment.
Can FootVIEW perform automatic center-of-pressure (COP) path smoothing and statistical outlier removal?
Yes—users may apply Savitzky-Golay filtering, median-based noise suppression, and customizable velocity/threshold gating to COP trajectories prior to kinetic parameter extraction.
Does the system support multi-subject concurrent acquisition in shared Wi-Fi environments?
Yes—each F-Scan GO unit operates on a unique SSID and channel assignment; up to eight units can be managed simultaneously via centralized FootVIEW Server deployment.
Are replacement insoles available with different material durometers for pediatric or high-arch applications?
Yes—Tekscan offers three durometer options (Soft, Medium, Firm) across all size ranges, validated for repeatability across 10,000+ loading cycles per insole.