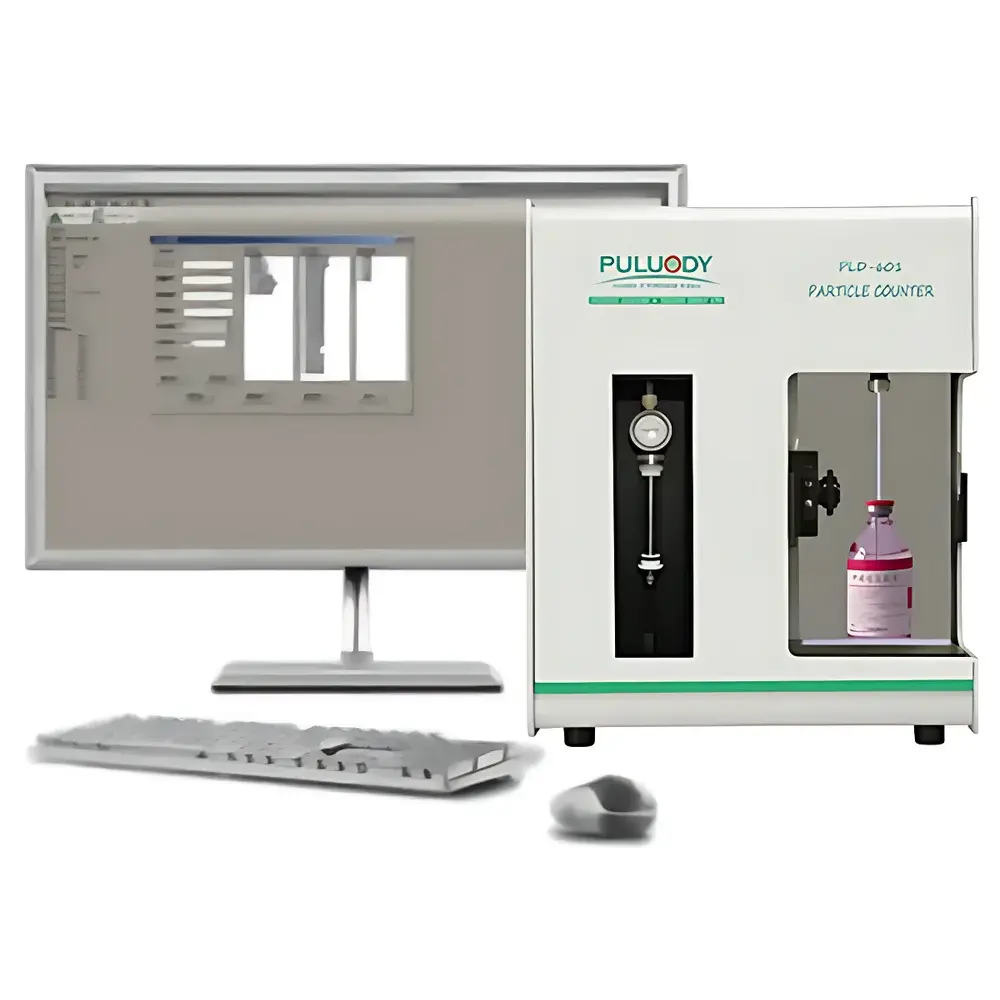
PULUODY PLD-601 Pharmaceutical Insoluble Particulate Analyzer for Injectable and Packaging Materials
| Brand | PULUODY |
|---|---|
| Origin | Shaanxi, China |
| Model | PLD-601 |
| Dispersion Method | Wet Dispersion |
| Instrument Type | Laboratory Laser-Based Light Obscuration (LO) Particle Counter |
| Measurement Range | 1–500 µm |
| Repeatability | 1000 particles/mL (2.5% coincidence error) |
| Measurement Time | 60 seconds |
Overview
The PULUODY PLD-601 Pharmaceutical Insoluble Particulate Analyzer is a laboratory-grade, benchtop light obscuration (LO) particle counter engineered for regulatory-compliant quantification and sizing of insoluble particulates in parenteral drug products, sterile raw materials, medical packaging components, and purified water systems. Unlike laser diffraction or dynamic light scattering instruments, the PLD-601 employs a dual-laser, narrow-beam optical detection principle based on ISO 21501-4 and USP , where particles suspended in a liquid medium interrupt a collimated laser beam, generating voltage pulses proportional to their cross-sectional area. This direct, real-time volumetric counting method ensures traceable, pharmacopeial-grade data for subvisible particle enumeration—critical for assessing injectable safety, filter validation, container-closure integrity, and process-related contamination. The instrument complies with compendial requirements for both routine QC release testing and investigative analysis across GMP, GLP, and R&D environments.
Key Features
- Dual-laser, narrow-beam optical sensor (8th-generation PULUODY proprietary design) delivering enhanced signal-to-noise ratio and minimized optical noise at low particle concentrations.
- Dual-precision fluidic control system: high-accuracy metering piston pump combined with ultra-stable electromagnetic flow regulation, ensuring volumetric sampling repeatability within ±1% and flow stability across 5–150 mL/min.
- 1000-channel high-resolution pulse-height analysis (PHA), enabling customizable binning from 0.01 µm upward—fully configurable to meet USP , EP 2.9.19, ChP 2020 Edition 0903, and JP 17 specifications.
- FDA 21 CFR Part 11-compliant software architecture with five-tier user permission levels, electronic signatures, full audit trail, automatic data backup/recovery, and power-fail recovery.
- Integrated temperature-controlled fluid path (0–80 °C) and ambient operating range (−15 to 50 °C), supporting analysis of viscous formulations, cold-chain biologics, and high-purity water streams.
- Calibration traceability to NIM Northwest Metrology Testing Center or Defense Science & Technology Commission Level-1 Particle Calibration Station No. 116 per JJG 1061, ISO 21501-4, and latex standard reference materials.
Sample Compatibility & Compliance
The PLD-601 is validated for use with a broad spectrum of pharmaceutical and biopharmaceutical liquids including: solution-type injections, sterile powders for reconstitution, concentrated injection solutions, sterile active pharmaceutical ingredients (APIs) for IV use, ophthalmic solutions, vaccines, Water for Injection (WFI), Purified Water (PW), medical packaging extracts (e.g., YBB00272004-2015), and infusion device leachates. It supports terminal filter efficiency evaluation per ISO 13408-2 and particulate removal rate assessment under controlled flow conditions. Regulatory alignment includes ChP 2020 Edition 0903, USP //, EP 10.0/9.0/8.0, BP 2019/2018, JP 17/16/15, WHO International Pharmacopoeia IV, and ISO 21510, ISO 11171, and ASTM D6786 for calibration and reporting consistency.
Software & Data Management
The V8.9 PC-based analytical software provides full instrument control, real-time pulse visualization, automated pass/fail reporting per user-defined thresholds, and multi-standard compliance templates. Data integrity safeguards include time-stamped audit logs, immutable electronic records, role-based access control, and encrypted local/cloud backups. All reports export to PDF, CSV, or XML formats with metadata embedding (operator ID, calibration status, environmental parameters). Software validation documentation (IQ/OQ/PQ protocols) and 21 CFR Part 11 implementation guides are provided as part of the delivery package.
Applications
- Pharmaceutical QC labs: Routine batch release testing of injectables per pharmacopeial monographs.
- Filter validation studies: Quantitative assessment of retention efficiency for 0.22 µm and 0.45 µm sterilizing-grade membranes.
- Container-closure system evaluation: Extractables and leachables screening from vials, stoppers, and IV bags.
- Water system monitoring: Particulate load trending in WFI, PW, and ultrapure water loops per ASTM D5127 and ISO 3696.
- Biomanufacturing support: Monitoring of cell culture media, buffer solutions, and final fill streams for process-related particulates.
- Medical device testing: Particulate shedding from catheters, syringes, and infusion sets per GB 8368 and ISO 8536-4.
FAQ
Does the PLD-601 comply with USP and ChP 2020 0903?
Yes—the optical configuration, calibration protocol, and software reporting modules are preconfigured to satisfy all mandatory requirements of both standards, including sensitivity verification, system suitability, and reporting of ≥10 µm and ≥25 µm particle counts per mL.
Can the instrument measure particles below 1 µm?
The standard configuration covers 1–500 µm; optional extended-range sensors (0.03–3000 µm) are available upon request and require separate validation per ISO 21501-4 Annex B.
Is third-party metrological certification included?
Each unit ships with a traceable calibration certificate issued by either the NIM Northwest Metrology Testing Center or the Defense STC Level-1 Particle Calibration Station No. 116, compliant with JJG 1061.
What maintenance is required for long-term accuracy?
Annual recalibration is recommended; daily system suitability checks using NIST-traceable latex standards (e.g., 10 µm and 25 µm) are required prior to regulated testing.
How is data integrity ensured during networked operation?
All network communication uses TLS 1.2 encryption; database writes are transaction-locked and journaled; audit trails cannot be edited or deleted without documented supervisor override.