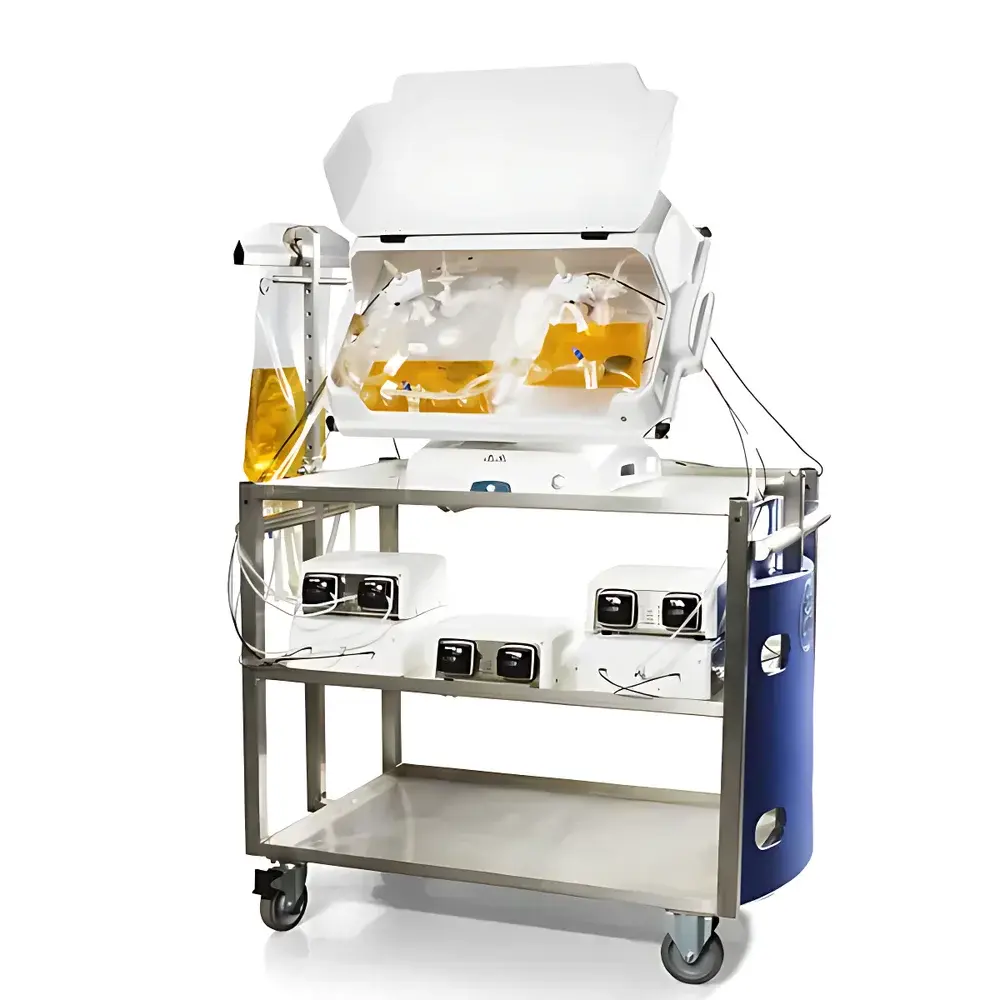
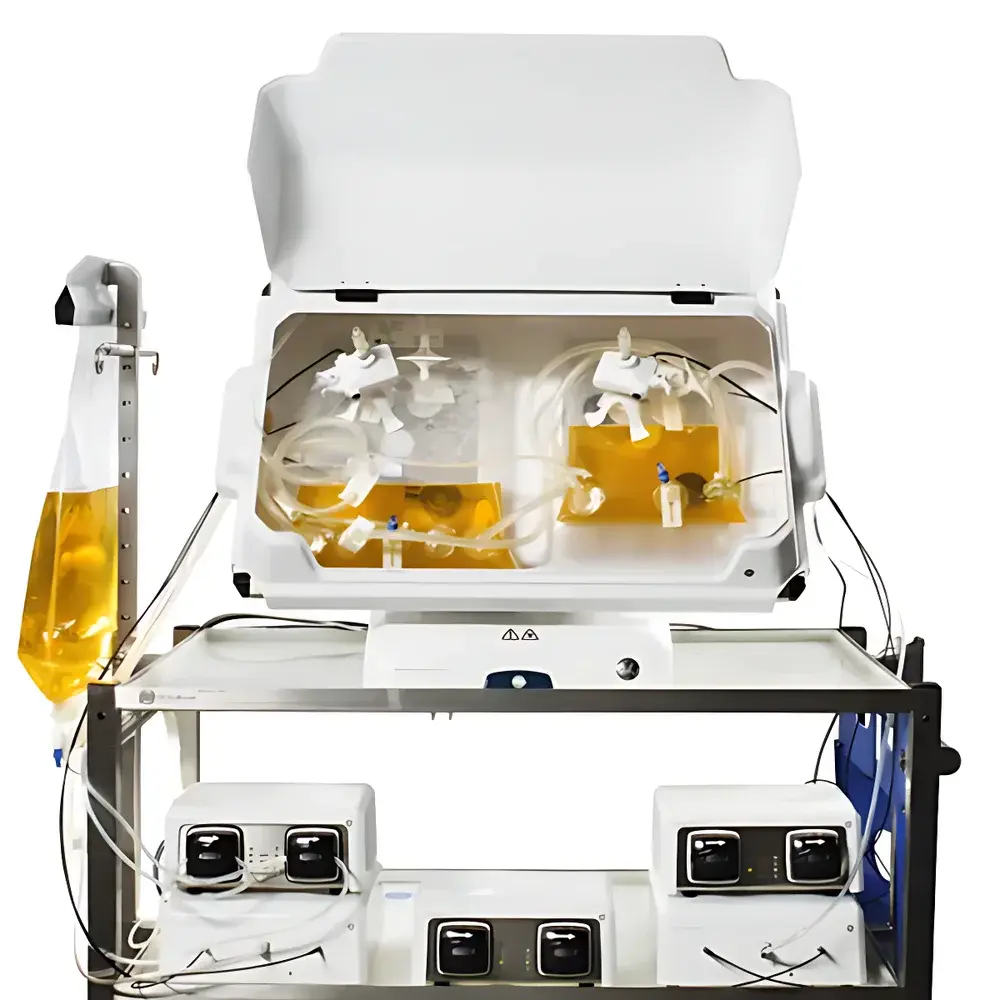
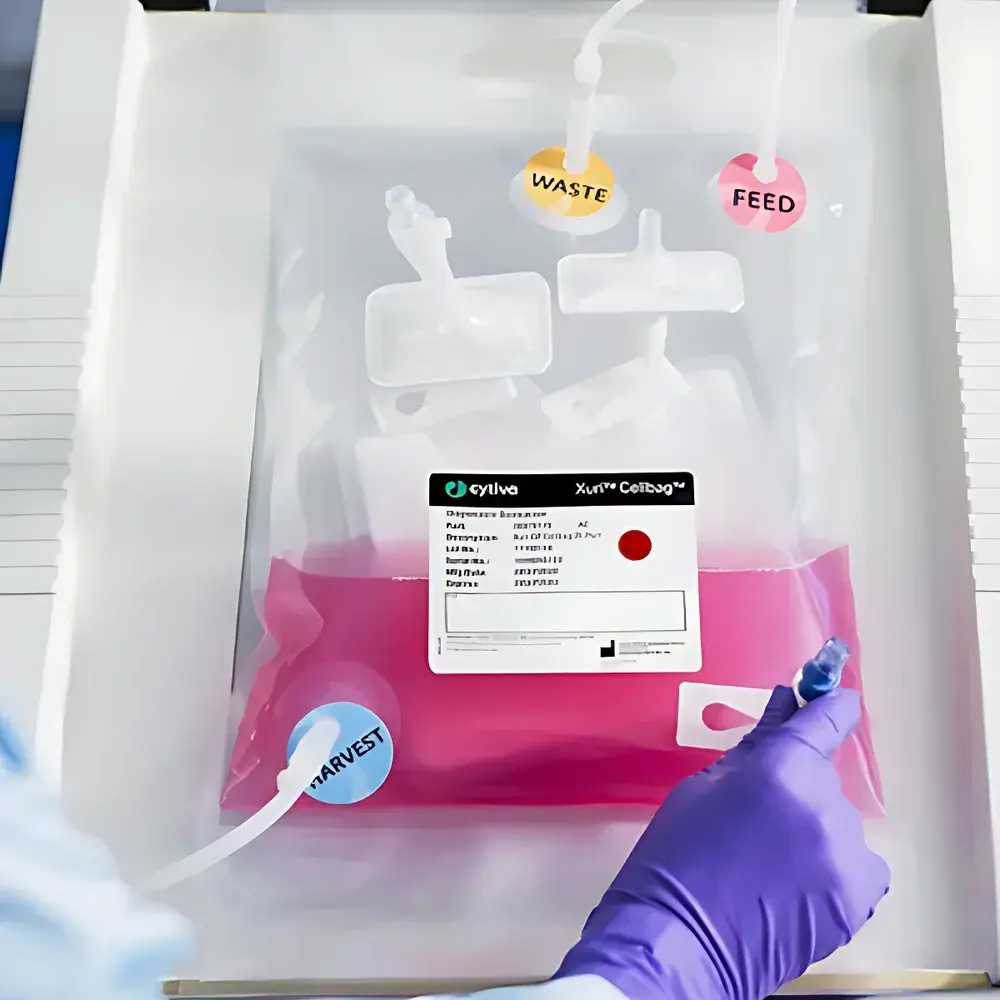
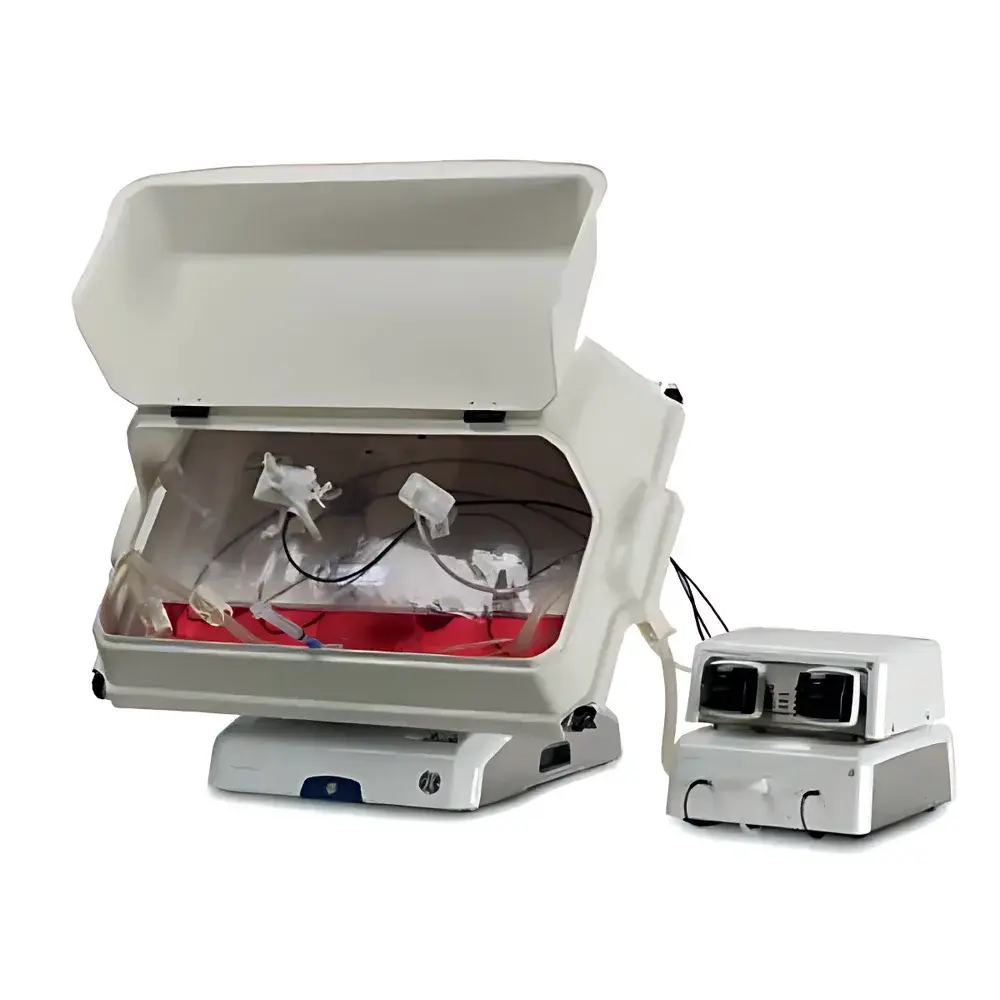
- All
- Favorite
- Popular
- Most rated
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | WAVE 20/50 |
| Maximum Working Volume | 25 L |
| Tray Sizes | 5 L / 10 L / 25 L |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | ÄKTA readyflux |
| Type | Single-Use Tangential Flow Filtration (TFF) System |
| Application Scope | Lab-Scale to Pilot-Scale Bioprocessing |
| Control Software | UNICORN 7.x or later |
| Compatibility | DeltaV™ DCS, ReadyToProcess Hollow-Fiber Cartridges, Xcellerex XDUO Single-Use Mixers, Third-Party Bag Mixers |
| Regulatory Alignment | Designed for GLP/GMP environments |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex™ X-50 |
| Pricing | Upon Request |
| Application Domain | Biopharmaceutical Cell Culture |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | UniFlux Tangential Flow Filtration System |
| Flow Rate Options | 10, 60, 120 L/min (400 L/min available on custom basis) |
| Vessel Capacity Range | 5–600 L (stainless steel or single-use mixing systems supported) |
| Automation Platform | UNICORN Software (21 CFR Part 11 compliant) |
| Application Scope | Microfiltration (e.g., cell harvest) and Ultrafiltration (e.g., protein concentration & diafiltration) |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Model | ReadyToProcess WAVE 25 |
| Maximum Working Volume | 25 L |
| Drive Mechanism | Motorized Rocking Platform with Adjustable Speed, Angle, and Motion Profile |
| Temperature Control | Integrated Heated Tray with Dual Independent PID Loops (Dual-Mode Operation) |
| Weight Measurement | Built-in Load Cells with Auto-Calibration Capability |
| Gas Transfer | Passive O₂/CO₂ Exchange via Sterile Vent Filters (Heated Exhaust Filter Included) |
| Data Storage | Dual Mirrored SSDs + UNICORN Database Integration |
| Communication Protocol | OPC UA Compliant |
| Compliance | Designed for GMP Environment |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ Spark™ Instrument |
| Sample Volume Range | 25–250 µL |
| Mixing Technology | NxGen™ Microfluidic Vortex Mixing |
| Sterility Design | BSC-Compatible, Aseptic Operation Ready |
| Chip Type | Single-Use, Gamma-Irradiated Cartridge |
| Scalability Pathway | Seamless Transition to NanoAssemblr™ Ignite™ and NxGen™ Production Platforms |
| Regulatory Context | Designed for GLP-compliant early-stage development, supports 21 CFR Part 11–ready software integration (optional) |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | Wave Cellbag |
| Sterilization Method | Gamma Irradiation |
| Material Compliance | USP Class VI |
| Regulatory Documentation | FDA-registered DMF on file |
| Intended Use | Research, Process Development, cGMP Manufacturing, Cell Therapy Production |
| Bag Configuration | Single-use, pre-sterilized, disposable bioprocess bag |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | iCELLis™ Nano Bioreactor System |
| Temperature Range | Double-jacketed with TCU – 20–40 °C |
| Surface Area Range (iCELLis™ 500+) | 60–500 m² |
| Surface Area Range (iCELLis™ Nano) | 0.53–4 m² |
| Configuration | Single-use, integrated fixed-bed bioreactor |
| Operating Modes | Batch and perfusion |
| Compliance | Designed to support GMP-compliant manufacturing |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ Blaze™ System |
| Volume Range | 1 L (Blaze™) / up to 10 L (Blaze+™) |
| System Architecture | Closed, single-use, GMP-ready process development platform |
| Core Technology | NxGen™ microfluidic mixing |
| Regulatory Alignment | Supports FDA 21 CFR Part 11–compliant data integrity, GLP/GMP process transfer readiness |
| Disposable Components | NxGen™ single-use chips (with/without inline dilution), sterile tubing kits for external vessel integration |
| Downstream Compatibility | Integrated TFF, sterile filtration, and fill-finish workflow support |
| Scalability Pathway | Direct CPP/CQA transfer from Blaze™ → Blaze+™ → NanoAssemblr™ Production System |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | NanoAssemblr™ Commercial Manufacturing System |
| Pricing | Upon Request |
| Flow Rate Range | 6–48 L/h (dependent on disposable flow path kit) |
| Brand | Cytiva |
|---|---|
| Origin | Imported |
| Manufacturer Type | Authorized Distributor |
| Membrane Area Compatibility | 0.1–2.5 m² (cassette & hollow-fiber modules) |
| Predefined Process Programs | UF Production, Normalized Water Permeability (NWP), Clean-in-Place (CIP), Water Rinse, Alkaline Hold |
| Regulatory Alignment | Designed to support GMP, FDA 21 CFR Part 11, and EU Annex 11 compliance requirements |
| Software Architecture | Embedded control with audit trail, user access levels, and electronic signature capability |
| System Flexibility | Modular configuration for lab-scale development, pilot-scale process validation, and small-batch cGMP manufacturing |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ GMP System |
| Flow Rate Range | 60–200 mL/min (3.6–12 L/h) |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | ÄKTA flux 6 |
| Configuration | Semi-automated TFF platform |
| Application Scope | Concentration, diafiltration, cell harvest & clarification |
| Membrane Compatibility | Hollow-fiber modules & cassette-type membrane packs |
| Operating Volume Range | 50 mL – 6 L |
| Control Interface | Integrated touchscreen with real-time parameter monitoring |
| Data Logging | Onboard process recording (time-stamped, user-ID traceable) |
| Regulatory Support | Compliant with FDA 21 CFR Part 11 audit trail requirements for GMP-relevant workflows |
| Service Framework | OptiRun lifecycle support program |
| Change Notification | Web-based Change Control Notification (CCN) service |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Xuri Cell Expansion System W25 |
| Price | Upon Request |
| Temperature Range | 15°C–32°C |
| Working Volume | 25 L |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | BioProcess Resin Mixer |
| Pricing | Upon Request |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ Ignite™ Instrument |
| Flow Rate Range | 20–200 mL/min |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | ÄKTA flux s |
| Application Scope | Research & Membrane Screening |
| Control Mode | Automated Endpoint Control, Constant Retentate Volume (CRV) Mode |
| User Interface | 7-inch High-Resolution Touchscreen with Real-Time Process Monitoring |
| Data Handling | Onboard Logging (CSV Export), Audit-Trail-Ready Event History |
| Compliance Context | Designed for GLP-compliant workflows |
| Brand | Cytiva |
|---|---|
| Origin | Manufactured in China under Cytiva’s global quality system |
| Manufacturer Type | Authorized Distributor |
| Model | Agile flux TFF Systems |
| Pricing | Available upon request |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | ÄKTA flux 6 |
| Application Scope | Process Development & Small-Scale Production |
| Control Mode | Automated Endpoint & Constant Retentate Volume (CRV) |
| Data Handling | Real-Time Logging with Audit-Trail-Capable Software |
| Regulatory Compliance | Designed for GMP Environments |
| Installation & Qualification | IQ/OQ Support Available by Certified Engineers |
| User Interface | Integrated Touchscreen HMI |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex XDR-50 MO |
| Application Domain | Microbial Fermentation |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | STF-IRc |
| Tube OD Capacity | Up to 22.2 mm |
| Automation Level | Fully Automated |
| Sterility Assurance | Validated Aseptic Connection without Biosafety Cabinet |
| Application Domain | Bioprocessing Fluid Transfer |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex XDR 50–2000 |
| Working Volumes | 50 L, 200 L, 500 L, 1000 L, 2000 L |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | Wave Mixer |
| Type | Sterile, Non-invasive Oscillatory Mixing System |
| Capacity | Up to 10 L |
| Mixing Time | ≤6.5 s for full homogenization |
| Drive Mechanism | Programmable Rocking Platform with Sinusoidal Motion Profile |
| Sterility Assurance | Designed for use with single-use bioprocess bags (e.g., Cytiva HYPERSTACK®, Xcellerex™ XDR bags) |
| Compliance | Compatible with ISO 13485–certified manufacturing environments and supports GMP-aligned process validation |
| Brand | Cytiva |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | GE ReadyToProcess WAVE 25 Bioreactor |
| Temperature Range | 6–60 °C |
| Working Volume Range | 10–2000 L |
| Number of Vessels | 1 |
| Construction Material | Stainless Steel |
| Sterilization Method | Single-Use Bioreactor System |
Show next