SurgiVet V9204 Veterinary Patient Monitor by Harvard Apparatus
| Brand | Harvard Apparatus |
|---|---|
| Origin | USA |
| Model | V9204 |
| Display | 10.4" color TFT touchscreen |
| ECG | 3-lead or 5-lead configurable |
| Blood Pressure | Non-invasive (NIBP) and invasive (IBP) monitoring |
| SpO₂ | Digital signal processing with Sensor Auto-Calibration (SAC) technology |
| Temperature | Dual-channel analog input |
| Respiration | Impedance-based |
| Weight | ≤ 5.4 kg (12 lbs) |
| Compliance | FDA-cleared for veterinary use, CE-marked, ISO 13485 certified |
Overview
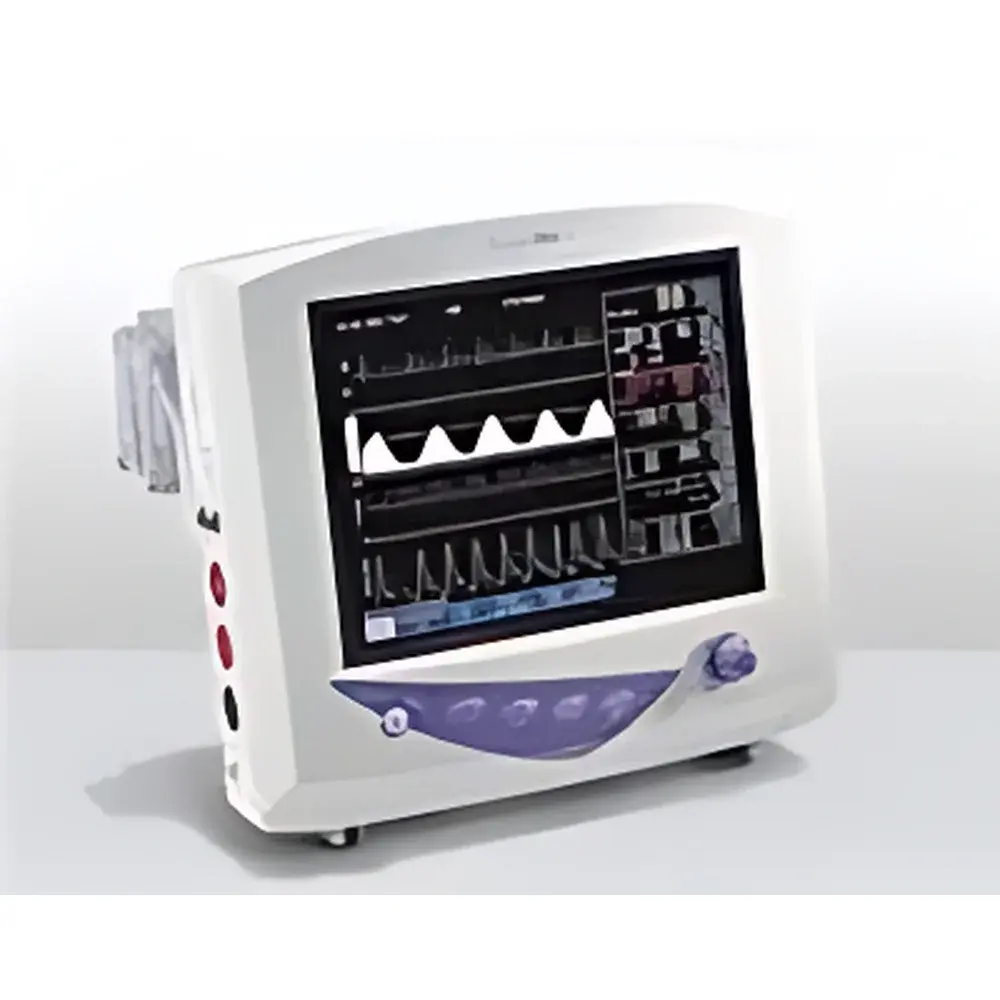
The SurgiVet V9204 Veterinary Patient Monitor is a portable, multi-parameter physiological monitoring system engineered for precision and reliability in preclinical research, surgical support, and critical care settings involving laboratory animals and companion species. Designed and manufactured under the quality management framework of Harvard Apparatus—a U.S.-based leader in life science instrumentation—the V9204 integrates core hemodynamic, respiratory, and metabolic parameters using clinically validated measurement principles. Its electrocardiographic acquisition employs standard lead configurations (3-lead or 5-lead selectable) with real-time R-wave detection and arrhythmia flagging. Blood pressure monitoring supports both oscillometric non-invasive measurement (NIBP) and high-fidelity invasive arterial pressure waveform analysis (IBP) via analog pressure transducer inputs. Oxygen saturation (SpO₂) utilizes digital photoplethysmography with Sensor Auto-Calibration (SAC), minimizing motion artifact and ensuring stable readings across small-animal models including rodents, rabbits, canines, and felines. Respiratory rate is derived from thoracic impedance changes, while dual-channel temperature monitoring accommodates rectal, esophageal, or surface probe configurations—critical for thermoregulation studies and anesthesia protocols.
Key Features
- 10.4-inch high-resolution color TFT display with capacitive touchscreen interface and intuitive rotary encoder navigation—optimized for gloved-hand operation in sterile environments
- Simultaneous real-time display of up to four waveforms (ECG, IBP/NIBP, SpO₂ plethysmogram, respiration) with adjustable timebase and amplitude scaling
- Integrated thermal printer for immediate hard-copy documentation of vital signs trends, alarm events, and snapshot reports—supporting GLP-compliant record retention
- Rechargeable lithium-ion battery providing ≥ 4 hours of continuous operation under typical multi-parameter acquisition conditions
- Rugged, lightweight chassis (≤ 5.4 kg) with integrated carrying handle and shock-absorbing rubberized corners—designed for transport between procedure rooms, vivariums, and mobile field units
- Configurable alarm thresholds with visual (flashing icons, color-coded status bars) and audible (multi-tone, user-adjustable volume) alerting—fully programmable per parameter and animal species
Sample Compatibility & Compliance
The V9204 is validated for use across a broad spectrum of vertebrate models—from murine (mouse, rat) and lagomorph (rabbit) subjects to larger species including swine, canine, and feline. Its scalable sensor interface architecture accepts industry-standard veterinary probes and transducers compliant with IEC 60601-1 (3rd ed.) and IEC 60601-2-49 (for patient monitors). The device bears CE marking under the EU Medical Device Regulation (MDR 2017/745) and is FDA 510(k)-cleared (K number on file) for veterinary diagnostic and monitoring applications. All firmware and calibration routines adhere to ISO 13485:2016 requirements, and data logging complies with ALPACO and AAALAC International guidelines for institutional animal care and use committees (IACUC).
Software & Data Management
Data acquisition and visualization are managed through embedded firmware supporting 128-second waveform memory buffer and trend storage for up to 72 hours of selected parameters at 1 Hz sampling. Export options include USB mass-storage mode for CSV-formatted trend logs and PDF snapshot reports—compatible with institutional LIMS and electronic lab notebook (ELN) platforms. While the V9204 does not feature network connectivity or cloud integration, its timestamped, audit-trail-enabled data output satisfies FDA 21 CFR Part 11 requirements when paired with validated local archiving systems. Firmware updates are delivered via secure USB drive and require administrative authentication to maintain configuration integrity.
Applications
- Anesthesia monitoring during acute surgical procedures in rodent stereotaxic setups and large-animal operating theaters
- Longitudinal cardiovascular phenotyping in genetically modified mouse models (e.g., hypertensive, arrhythmic, or heart failure strains)
- Toxicology studies requiring continuous hemodynamic assessment per OECD Test Guideline 457 (hERG assay support)
- Preclinical efficacy evaluation of novel vasoactive or respiratory therapeutics under GLP conditions
- Vivarium-based post-operative recovery surveillance with customizable alarm profiles for species-specific normative ranges
FAQ
Is the V9204 compatible with third-party pressure transducers and SpO₂ sensors?
Yes—provided they conform to standard analog output specifications (e.g., 0–5 V DC for IBP, 0–5 V or 4–20 mA for temperature) and FDA-cleared veterinary SpO₂ probe form factors.
Does the device support pediatric or neonatal animal monitoring?
It is validated for use in animals weighing ≥ 200 g; signal sensitivity and alarm algorithms may require manual adjustment for neonatal rodent applications.
Can waveform data be streamed in real time to external acquisition systems?
No—real-time analog output ports are not included; only buffered export via USB is supported.
What is the recommended calibration frequency for NIBP and SpO₂ modules?
Harvard Apparatus recommends annual verification against traceable standards; NIBP cuff calibration requires hydrostatic column validation, while SpO₂ accuracy should be verified using calibrated blood gas analyzers per ISO 80601-2-61.
Is the thermal printer replaceable or serviceable in the field?
The integrated thermal print mechanism is a sealed module; replacement requires authorized service center intervention and firmware reinitialization.