Advion Interchim Scientific OPSI Open Port Sampling Interface
| Brand | Advion |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | OPSI Open Port Sampling Interface |
| Pricing | Upon Request |
Overview
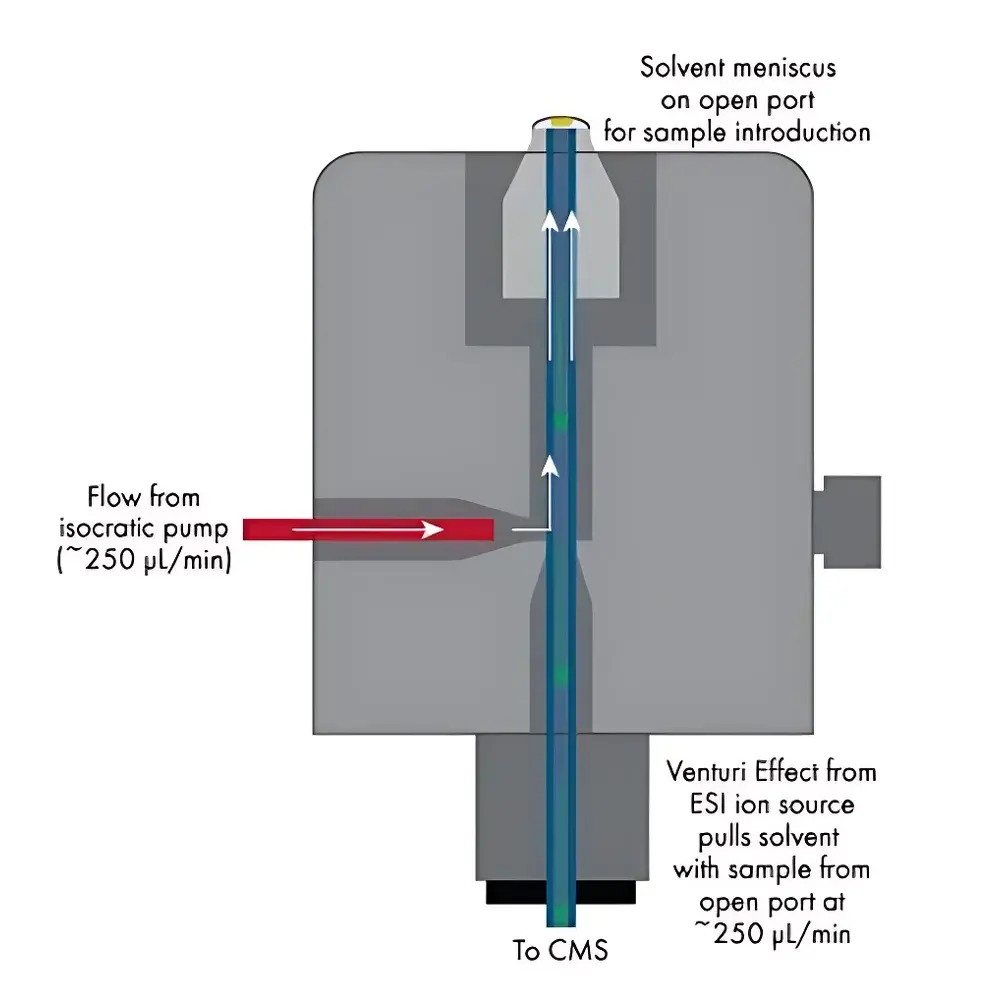
The Advion Interchim Scientific OPSI (Open Port Sampling Interface) is an ambient ionization source engineered for direct, real-time mass spectrometric analysis of solid, liquid, and surface-bound analytes under atmospheric pressure. Developed from foundational research at Oak Ridge National Laboratory by Drs. Gary Van Berkel and Vilmos Kertesz, the OPSI integrates a low-flow solvent delivery system with a Venturi-assisted aspiration mechanism to enable rapid desorption and electrospray-like ionization without vacuum lockout or sample introduction hardware modification. When coupled with the expression® CMS — a compact, benchtop single-quadrupole mass spectrometer — the OPSI functions as a fully integrated open-port interface that transports analyte ions directly into the instrument’s ion source region via solvent-mediated extraction. This design eliminates the need for chromatographic separation, matrix application, or vacuum-compatible sample loading, supporting sub-second detection of small molecules, lipids, nucleotides, peptides, and synthetic polymers directly from untreated surfaces.
Key Features
- Atmospheric-pressure ambient ionization compatible with standard electrospray ionization (ESI) optics and vacuum interfaces
- Low-flow solvent delivery (typically 0.1–2 µL/min) enabling minimal sample consumption and reduced chemical noise
- Venturi-driven aspiration ensures stable, pulse-free analyte transfer independent of sample geometry or volatility
- No high-voltage electrodes in contact with the sample — reduces electrochemical degradation and improves reproducibility for redox-sensitive compounds
- Modular mechanical integration with expression CMS; requires no firmware or software customization
- Compatible with both positive- and negative-ion mode acquisition across m/z 50–2000
Sample Compatibility & Compliance
The OPSI supports direct analysis of untreated solids (e.g., tablets, plant tissues, forensic swabs), liquids (including viscous oils and biological fluids), and micro-regional surfaces (e.g., TLC plates, tissue sections, polymer films). Its ambient operation aligns with ASTM E3179-21 guidelines for ambient desorption/ionization methods and facilitates compliance with GLP/GMP documentation requirements when paired with expression CMS’s audit-trail-enabled data acquisition software. While not a standalone regulated device, the OPSI–expression CMS configuration has been validated in laboratories adhering to FDA 21 CFR Part 11 for electronic records and signatures during method development for pharmaceutical impurity profiling and food contaminant screening.
Software & Data Management
Operation is fully controlled through the expression CMS’s proprietary MassExpress™ software, which provides synchronized solvent flow control, real-time spectral preview, and automated peak integration. All acquisition parameters — including solvent composition, flow rate, capillary temperature, and detector gain — are stored with raw data files in vendor-native .raw format. The software supports batch processing, spectral library matching (NIST, Wiley, custom libraries), and export to common formats (.mzML, .csv) for third-party chemometric analysis. Audit trails record user actions, parameter changes, and instrument state transitions, satisfying traceability requirements for regulated environments.
Applications
- Pharmaceutical R&D: Rapid identification of active pharmaceutical ingredients (APIs), excipients, and degradation products on tablet surfaces or in dissolution media
- Food safety: On-site screening for pesticides, mycotoxins, and adulterants in fruit skins, grain surfaces, or processed dairy matrices
- Environmental monitoring: Direct detection of PAHs, PCBs, and flame retardants on soil particles, filter membranes, or sediment cross-sections
- Forensic toxicology: In situ analysis of drugs of abuse on banknotes, clothing fibers, or latent fingerprint residues
- Lipidomics & metabolomics: Spatially resolved profiling of phospholipids and glycerolipids from frozen tissue sections without matrix application
FAQ
Does the OPSI require vacuum interruption during sample introduction?
No — the interface operates continuously at atmospheric pressure; no venting or pump-down cycles are needed.
Can the OPSI be used with other mass spectrometers besides expression CMS?
Yes, with appropriate mechanical and electrical interfacing; however, full plug-and-play functionality and optimized ion transmission are guaranteed only with expression CMS systems.
Is matrix application required for solid sample analysis?
No — the solvent-mediated desorption process eliminates the need for MALDI-type matrices.
What solvents are compatible with the OPSI?
Common ESI solvents including methanol, acetonitrile, water, and mixtures containing up to 0.1% formic acid or ammonium acetate; viscosity and surface tension must remain within operational limits for stable microflow delivery.
How is calibration performed?
Standard external calibration using infused tuning compounds (e.g., sodium formate clusters) is recommended prior to each analytical session; internal standards may be co-infused for quantitative workflows.