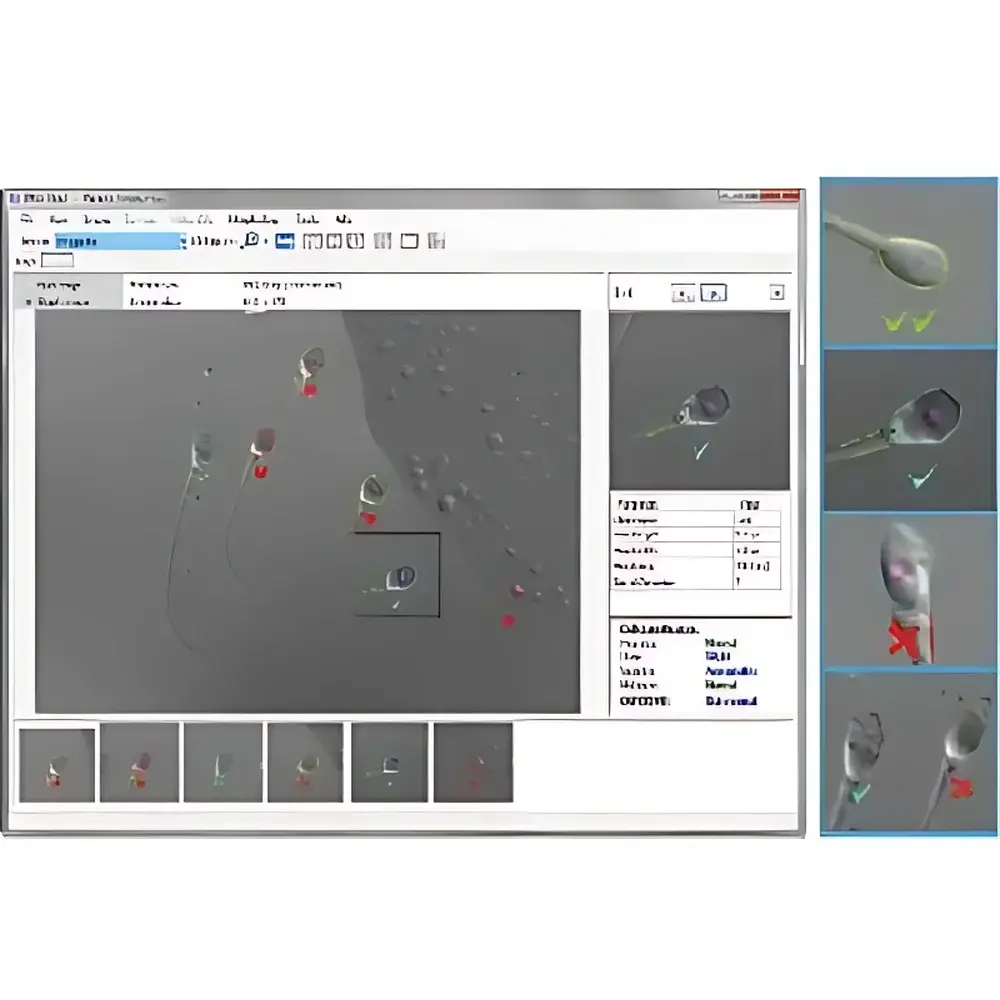
Hamilton Thorne IMSI Strict Automated Sperm Morphology Analysis Software
| Brand | Hamilton Thorne |
|---|---|
| Origin | USA |
| Model | IMSI Strict |
| Application | Clinical Embryology, ART Laboratories |
| Compatibility | Leica, Nikon, Olympus, Zeiss Inverted Microscopes |
| Camera Support | Universal USB 3.0 / GigE / Camera Link Interface |
| Analysis Mode | Real-time Live Imaging |
| Output | PDF/CSV Reports with Audit Trail |
| Regulatory Context | Designed for ISO 15189-compliant IVF labs |
Overview
The Hamilton Thorne IMSI Strict Automated Sperm Morphology Analysis Software is a CE-marked, FDA-listed Class I medical device software platform engineered for high-precision morphological assessment of human spermatozoa in assisted reproductive technology (ART) settings. Built upon the clinical standard of Intracytoplasmic Morphologically-selected Sperm Injection (IMSI), the software implements quantitative digital image analysis based on strict Kruger criteria (WHO 5th Edition, 2010) and extended ultrastructural parameters—including nuclear contour regularity, acrosomal integrity, midpiece symmetry, and flagellar morphology—at magnifications ≥6000× (achieved via high-NA oil immersion objectives and digital zoom calibration). Unlike conventional ICSI selection methods relying on subjective visual grading, IMSI Strict applies deterministic pixel-level segmentation algorithms to extract >27 morphometric features per sperm cell, enabling objective, reproducible, and traceable selection decisions aligned with ESHRE/ASRM best practice guidelines.
Key Features
- Real-time morphometric analysis during live microscopy—no frame capture delay; latency <120 ms from image acquisition to classification
- Multi-parameter validation engine: simultaneous evaluation of head area (µm²), head ellipticity ratio, acrosome-to-head area ratio (%), midpiece width uniformity (CV <8%), and tail bending angle distribution
- Color-coded morphological grading: green (normomorphic), yellow (borderline), red (teratogenic)—visually mapped onto live video feed with sub-pixel overlay registration
- Open architecture API supporting integration with laboratory information management systems (LIMS) and electronic embryology records (e.g., Veeva Vault, Medidata Rave)
- Calibration-certified optical path compensation: auto-corrects for objective-specific magnification drift, tube lens aberration, and sensor vignetting across microscope platforms
- Embedded DICOM-SR (Structured Reporting) export module compliant with IHE-ART technical framework for longitudinal patient data archiving
Sample Compatibility & Compliance
The IMSI Strict software processes unstained or Papanicolaou-stained sperm smears prepared on standard glass slides (76 × 26 mm), as well as wet-mount preparations under phase contrast or differential interference contrast (DIC) illumination. It is validated for use with motile and immotile sperm populations, including cryopreserved-thawed specimens. The system meets ISO 13485:2016 requirements for medical device software lifecycle management and incorporates built-in audit trail functionality per FDA 21 CFR Part 11 Annex 11 (EU GMP) standards—recording user ID, timestamp, parameter adjustments, and final selection rationale for every analyzed field. All morphometric thresholds are configurable within clinically validated ranges defined by the Andrology Laboratory Manual (ALM) 4th Edition.
Software & Data Management
IMSI Strict operates on Windows 10/11 64-bit workstations with NVIDIA Quadro RTX 4000-class GPU acceleration. Data handling follows HIPAA-compliant encryption-at-rest (AES-256) and TLS 1.3 encrypted transmission protocols. Each analysis session generates a timestamped, digitally signed report containing raw image thumbnails, feature scatter plots, statistical summaries (mean ± SD, % normal forms), and operator-annotated notes. Export formats include PDF/A-2b (archival), CSV (for statistical modeling in R/Python), and HL7 CDA Level 3 documents for EMR interoperability. Version-controlled software updates are distributed via secure vendor portal with full change-log documentation and re-validation summaries.
Applications
- Clinical IMSI procedure support in IVF-ICSI cycles for male factor infertility patients with prior fertilization failure or recurrent implantation failure
- Research-grade sperm phenotyping in andrology studies investigating environmental toxin exposure, varicocele impact, or epigenetic biomarker correlation
- Quality assurance auditing of embryologist selection consistency across multi-site fertility networks
- Training module for embryology fellows using annotated case libraries with expert-reviewed ground-truth morphology labels
- Longitudinal cohort analysis in prospective ART outcome studies requiring standardized morphometric endpoints
FAQ
Does IMSI Strict require proprietary hardware?
No—it is a software-only solution designed for integration with existing inverted microscopes and scientific cameras meeting minimum resolution (≥5 MP) and frame rate (≥30 fps) specifications.
Can the software be used for non-human sperm analysis?
While optimized for human sperm morphology per WHO criteria, the underlying algorithmic framework supports custom training sets for bovine, murine, or porcine models under research-use-only (RUO) licensing.
How is measurement traceability ensured across different microscope setups?
Each installation undergoes optical calibration using NIST-traceable micrometer slide standards; calibration profiles are stored per microscope ID and automatically applied during analysis.
Is remote support and software update delivery available internationally?
Yes—Hamilton Thorne provides secure cloud-based update deployment and remote desktop-assisted troubleshooting compliant with GDPR and local data sovereignty regulations.
What validation documentation is provided upon installation?
Customers receive a Site Acceptance Test (SAT) report, IQ/OQ protocol templates, and a Software Validation Summary aligned with ISO/IEC 17025:2017 for testing laboratories.