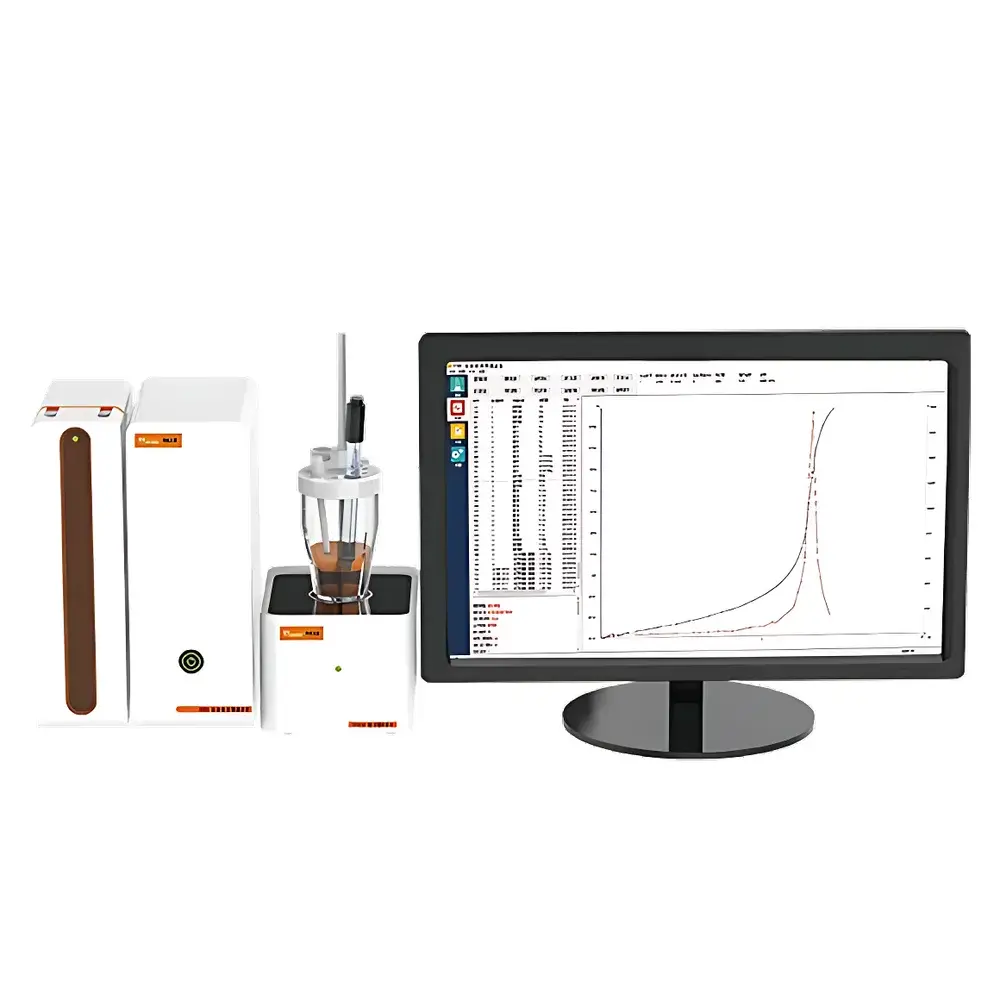
Hanon T910 Fully Automated Fatty Acid Value Titrator
| Brand | Hanon |
|---|---|
| Origin | Shandong, China |
| Model | T910 |
| Instrument Type | Universal Potentiometric Titrator |
| Application Scope | General-purpose |
| Measurement Range | 0–300 mg KOH/100 g dry matter |
| mV Range | −2000.0 to +2000.0 mV |
| pH Range | −20.000 to +20.000 pH |
| Electrode Readout Accuracy | RSD ≤ 0.3% |
| Resolution | 0.01 mg KOH/100 g dry matter |
| Titration Resolution | 1/1,500,000 |
| Temperature Range | −5 to 120 °C |
| Temp. Accuracy | ±0.1 °C |
| Titration Tube Options | 1 mL / 5 mL / 10 mL / 25 mL |
| Refill Time (100% fill) | 16 s |
| Communication Interfaces | Dual USB, RS-232, Ethernet, CAN bus |
| Electrode Ports | mV/pH input, reference electrode port, PT1000 temperature probe port |
| Audit Trail | FDA 21 CFR Part 11 compliant |
| Electronic Signature | Supported |
| Data Integrity | Immutable export with checksum verification |
| User Management | Role-based, multi-level permissions |
| Sample Handling | Standalone station or optional auto-samplers (16×100 mL, 12×250 mL, 18×50 mL) |
Overview
The Hanon T910 Fully Automated Fatty Acid Value Titrator is a purpose-engineered potentiometric titration system designed specifically for the quantitative determination of free fatty acid content in stored cereal grains—including wheat, corn, and rice—expressed as milligrams of potassium hydroxide required to neutralize free fatty acids per 100 grams of dry matter (mg KOH/100 g dry matter). Unlike classical endpoint detection using phenolphthalein indicator—which suffers from subjective visual interpretation, poor reproducibility, and inter-operator variability—the T910 employs a high-stability pH/mV sensing architecture coupled with dynamic endpoint recognition algorithms. It implements the electrochemical principle of potentiometric titration, where the inflection point of the titration curve (dE/dV vs. V) is precisely identified via second-derivative analysis or fixed potential threshold methods. This ensures objective, operator-independent results aligned with internationally referenced grain quality assessment protocols, including GB/T 20570–2015 (China), ISO 660:2020 (Animal and vegetable fats and oils — Determination of acid value and acidity), and AOAC Official Method 940.28 (Fatty Acids in Cereal Grains).
Key Features
- Pre-loaded, validated titration methods for grain-specific fatty acid value determination—fully compliant with national and international grain storage quality standards.
- Integrated titration burette housed within the instrument chassis, accessible via transparent observation window; eliminates open-burette handling and minimizes reagent exposure risk.
- Selectably configurable burettes (1 mL, 5 mL, 10 mL, 25 mL) matched to sample matrix and expected acid value range—enabling optimal volumetric resolution and reduced reagent consumption.
- Dual-mode stirring: selectable magnetic stirring (T9602A module) or overhead mechanical stirring (T9602 module) for heterogeneous grain extracts and viscous suspensions.
- Multi-stage self-diagnostic routine at startup and during operation—including electrode impedance check, liquid path integrity verification, and burette piston seal validation—to prevent erroneous titrations.
- Modular expansion capability: supports optional add-ons including wireless control (T9605), spray rinsing (T9606), automated lid actuation (T9607), and high-capacity auto-samplers (T9616 series).
- Real-time graphical display of titration curves (E vs. V), derivative plots (dE/dV vs. V), and endpoint confirmation markers—facilitating method development and troubleshooting.
Sample Compatibility & Compliance
The T910 accommodates solid, semi-solid, and liquid samples following standardized extraction procedures—typically ethanol–toluene solvent mixtures per GB/T 20570 or ISO 660. Its wide pH (−20.000 to +20.000) and mV (−2000.0 to +2000.0) measurement ranges support both strong-acid and weak-acid systems, while its ±0.003 pH accuracy and 0.1 mV resolution ensure reliable detection of subtle endpoint transitions. The system meets GLP and GMP operational requirements through full traceability: all user actions, method parameters, raw sensor data, titration curves, and result calculations are timestamped, digitally signed, and stored with cryptographic hashing. Audit trail records comply with FDA 21 CFR Part 11 Subpart B for electronic records and signatures—including role-based access control, immutable archival, and exportable PDF/A-2 reports with embedded digital signatures.
Software & Data Management
The embedded operating system features a dedicated titration software suite supporting six operational modes: MEAS (pH/mV/temperature monitoring), CAL (multi-point pH calibration), SET (fixed-endpoint titration), MET (equivalence-point titration), DET (dynamic endpoint tracking), and DOS (predefined volume dosing). Users may create, edit, and store custom titration sequences—including conditional logic (e.g., “if pH < 4.5, apply alternative titrant flow rate”), multi-step extraction integration, and post-titration cleaning cycles. Data export options include CSV (raw sensor logs), PDF (formatted reports with metadata), and XML (structured audit trail). Network connectivity via Ethernet or CAN bus enables centralized fleet management, remote diagnostics, and integration into LIMS environments using standard OPC UA or HTTP REST APIs.
Applications
- Grain quality surveillance in national reserve depots and commercial silos—tracking progressive lipolysis during long-term storage.
- QC/QA testing in flour mills and breakfast cereal manufacturers—verifying raw material acceptability prior to processing.
- Research applications in post-harvest physiology laboratories—correlating fatty acid value with moisture content, temperature history, and insect infestation metrics.
- Regulatory compliance testing for export certification—supporting documentation under SPS (Sanitary and Phytosanitary) measures and phytosanitary certificates.
- Method transfer and validation studies—leveraging the system’s reproducibility (RSD ≤ 0.3%) and traceable calibration protocols.
FAQ
What standards does the T910 support for fatty acid value determination?
The instrument implements methods aligned with GB/T 20570–2015, ISO 660:2020, and AOAC 940.28. Pre-configured workflows include solvent selection, titrant concentration, endpoint criteria, and unit conversion—all validated against certified reference materials.
Can the T910 be integrated into an existing LIMS or ELN platform?
Yes. Through native Ethernet and CAN bus interfaces, the system supports OPC UA server functionality and HTTP-based API endpoints for bidirectional data exchange—including sample ID push, method initiation, and structured result ingestion.
Is temperature compensation applied during titration?
Yes. A PT1000-grade temperature probe is integrated into each titration vessel, enabling real-time temperature correction of electrode response and automatic adjustment of titrant concentration based on thermal expansion coefficients.
How is data integrity ensured during long-term deployment?
All analytical sessions generate cryptographically signed, time-stamped records stored in a write-once internal database. Exported reports include SHA-256 checksums, digital signatures, and full audit trail lineage—meeting ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) principles.
What maintenance routines are recommended for sustained accuracy?
Daily: electrode hydration check and junction cleaning; Weekly: burette piston lubrication and leak test; Quarterly: full system performance verification using NIST-traceable pH and mV standards; Annually: factory recalibration with documented uncertainty budgets.