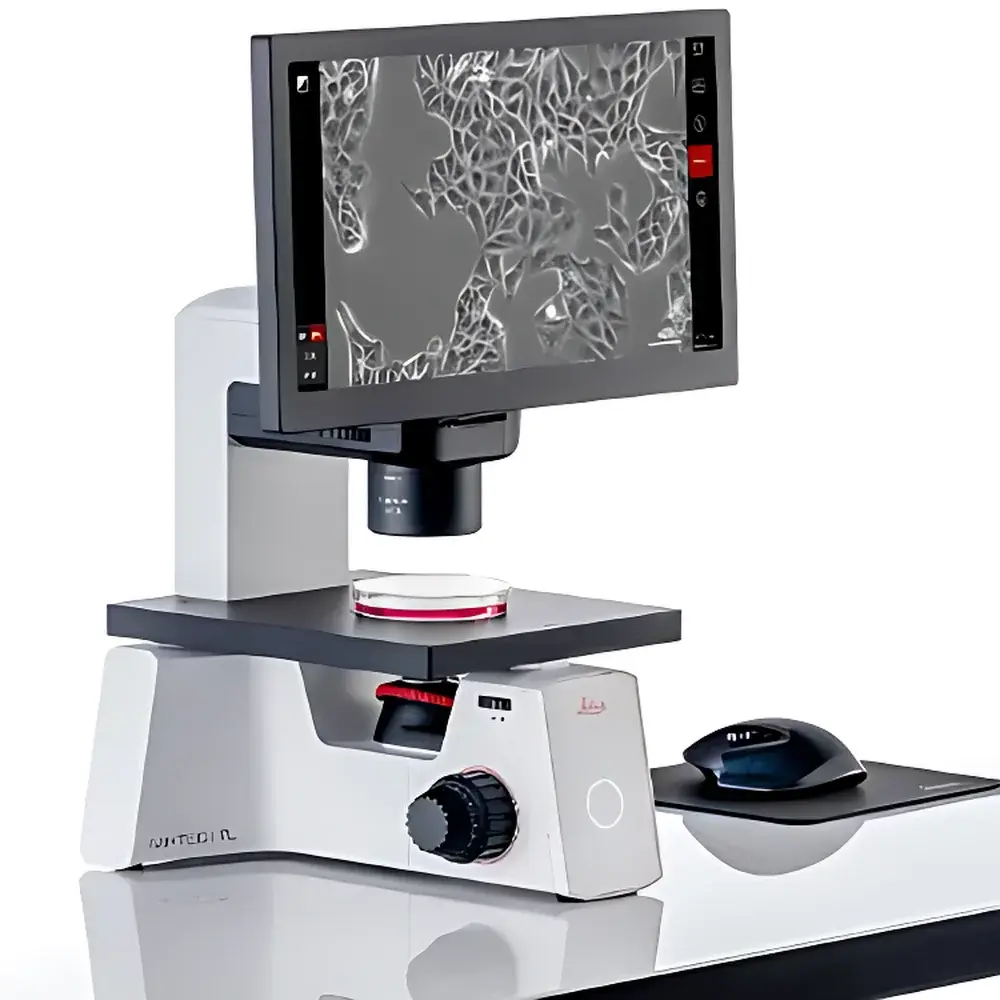
Leica Mateo TL Digital Inverted Microscope
| Brand | Leica |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin Classification | Domestic (China) |
| Model | Mateo TL |
| Instrument Category | Inverted Microscope |
| Pricing | Upon Request |
Overview
The Leica Mateo TL is a purpose-engineered digital inverted microscope designed for routine, high-throughput cell culture monitoring in academic, biopharmaceutical, and contract research organization (CRO) laboratories. Operating on standard brightfield and phase contrast optical principles, the Mateo TL eliminates reliance on eyepieces—delivering real-time, high-fidelity imaging directly to a calibrated display. Its optical path integrates an integrated CMOS sensor, fixed magnification objectives (typically 4×, 10×, 20×, and 40×), and LED-based Köhler illumination optimized for low phototoxicity and thermal stability during extended observation. Unlike traditional inverted microscopes requiring manual focus calibration or subjective confluence estimation, the Mateo TL embeds a validated confluence quantification algorithm based on pixel-level segmentation of adherent cell monolayers. This enables objective, operator-independent assessment of cell coverage percentage—critical for standardizing passaging decisions, transfection timing, and assay initiation across multi-user environments.
Key Features
- Digital-only workflow: No eyepieces required—full image acquisition, annotation, and export via touchscreen interface or external monitor.
- Integrated confluence analysis module: Automatically calculates % confluence using adaptive thresholding and morphological filtering; results are timestamped, user-tagged, and exportable as CSV or PDF reports.
- Intuitive phase contrast assistant: Guides users through condenser alignment and phase ring selection via step-by-step on-screen prompts—eliminating setup variability between novice and experienced operators.
- Light memory function: Stores illumination intensity and camera exposure settings per objective magnification—reducing re-optimization time between acquisitions.
- Benchtop-compatible design: Compact footprint (W × D × H ≈ 320 × 410 × 390 mm) and low-height base allow seamless integration into standard Class II biosafety cabinets and laminar flow hoods without compromising sterility or ergonomics.
- Wireless image sharing: Supports direct Wi-Fi transmission of still images and time-lapse sequences to iOS/Android devices for rapid peer review or documentation in electronic lab notebooks (ELNs).
Sample Compatibility & Compliance
The Mateo TL accommodates standard tissue-culture vessels including 6–96-well plates, T-flasks (T25 to T225), Petri dishes (35–150 mm), and chamber slides—without mechanical stage modification. Its working distance (≥ 35 mm at 10×) ensures unobstructed access to vessels under sterile hood conditions. All optical components comply with ISO 10934-1 (microscope nomenclature) and ISO 8578 (photomicrography accuracy). For regulated environments, the system supports IQ/OQ documentation packages aligned with ASTM E2500-13 (verification of laboratory equipment) and FDA 21 CFR Part 11 requirements when paired with Leica’s certified service team. Audit trails, user access logs, and metadata embedding (e.g., date/time, objective, confluence value, operator ID) are enabled by default in compliance with GLP and GMP-aligned workflows.
Software & Data Management
Leica Mateo TL operates on Leica Application Suite (LAS) X Lite—a lightweight, Windows-based platform supporting live imaging, batch capture, region-of-interest (ROI) marking, and automated confluence reporting. Raw image data is saved in TIFF format with embedded EXIF metadata (objective ID, exposure time, illumination intensity, software version). Confluence reports include statistical summaries (mean, SD, min/max across multiple fields of view) and optional overlay masks highlighting segmented cell regions. Data export supports DICOM, CSV, and PDF formats; integration with LabVantage, Benchling, and Veeva Vault is achievable via standardized API hooks. Software validation documentation (including installation qualification scripts and performance verification test protocols) is provided upon request for internal QA/QC departments.
Applications
- Cell culture quality control: Standardized confluence assessment prior to subculturing, cryopreservation, or differentiation induction.
- Transfection optimization: Timing nucleic acid delivery based on reproducible confluence thresholds (e.g., 70–80% for HEK293T, 50–60% for primary neurons).
- Bioprocess monitoring: Daily viability and morphology tracking in upstream biomanufacturing (e.g., CHO cell expansion in wave bags or bioreactors).
- Stem cell maintenance: Objective evaluation of colony density and edge uniformity in feeder-free pluripotent cultures.
- Educational labs: Low-barrier entry for undergraduate teaching modules on cell biology, cytotoxicity screening, and basic microscopy principles.
FAQ
Does the Mateo TL support fluorescence imaging?
No—the Mateo TL is configured exclusively for transmitted light modalities (brightfield and phase contrast). Fluorescence capability requires upgrade to the Leica DMi8 or DMI6 platform with appropriate filter cubes and excitation sources.
Can confluence measurements be validated against manual counting methods?
Yes—Leica provides a technical note (TN-MATEO-CL-01) detailing correlation studies against hemocytometer counts and automated image analysis tools (e.g., ImageJ with Trainable Weka Segmentation), demonstrating R² ≥ 0.92 across five common adherent lines (HeLa, A549, MCF-7, NIH/3T3, HUVEC).
Is remote operation supported?
Local network control is supported via VNC or RDP; however, cloud-based remote access is not natively implemented due to data governance policies governing sensitive biological image data.
What service plans include IQ/OQ documentation?
IQ/OQ execution and report generation are available as add-on services under Leica’s GoldCare or PlatinumCare extended warranty contracts—performed by factory-trained field service engineers with ISO/IEC 17025-accredited calibration certificates.
How frequently does the confluence algorithm require recalibration?
The algorithm is factory-validated and does not require user recalibration; periodic verification (e.g., quarterly) using Leica’s reference cell culture slide (Cat. No. 123456789) is recommended for labs operating under ISO 13485 or GxP frameworks.