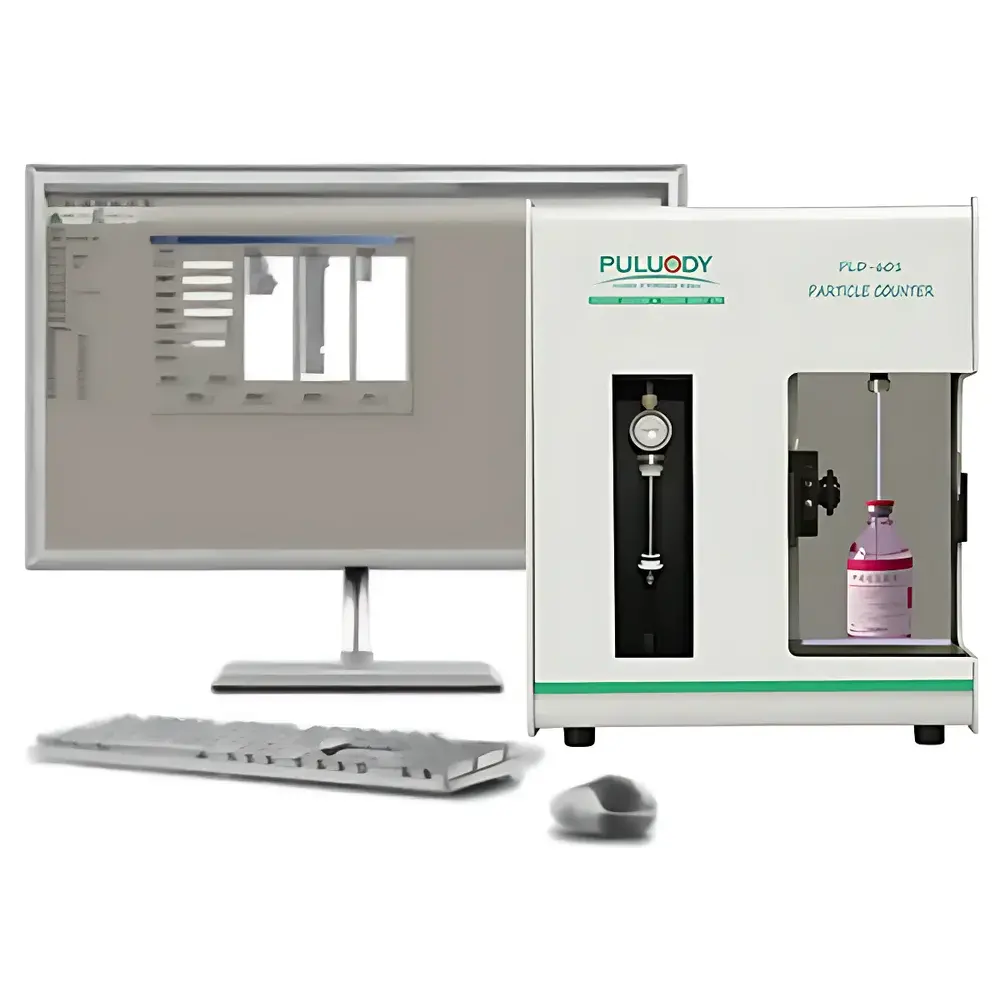
PULUODY PLD-601 Pharmaceutical Particulate Analyzer for Injectable Solutions and Medical Devices
| Brand | PULUODY |
|---|---|
| Origin | Shaanxi, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | PLD-601 |
| Price | USD 35,200 (FOB) |
| Dispersion Method | Wet Dispersion |
| Instrument Type | Benchtop Laboratory Laser Particle Counter |
| Measurement Range | 1–500 µm (0.03–3000 µm available upon configuration) |
| Repeatability | ≤2.5% coincidence error at 1000 particles/mL |
| Measurement Time | ≤60 seconds per sample |
Overview
The PULUODY PLD-601 Pharmaceutical Particulate Analyzer is a benchtop, regulatory-compliant laser-based particle counter engineered specifically for quantitative analysis of insoluble particulate matter in parenteral drug products, medical devices, and high-purity process liquids. It operates on the principle of light obscuration (also known as the optical blockage or resistive pulse method), where particles suspended in a liquid medium pass through a precisely defined sensing zone illuminated by dual narrow-beam lasers. Each particle generates a transient light extinction signal proportional to its projected area, enabling real-time sizing and counting across 1000 user-configurable channels with resolution down to 0.01 µm. Designed in strict alignment with pharmacopoeial requirements—including Chinese Pharmacopoeia (ChP) 2020 Edition General Chapter 0903, USP , , and , EP 10.0, BP 2019, and JP 17—the PLD-601 delivers traceable, auditable, and statistically robust data essential for quality control, filter validation, extractables/leachables assessment, and GMP-compliant release testing.
Key Features
- Dual eighth-generation narrow-beam laser sensor architecture for enhanced signal-to-noise ratio and reduced optical interference in turbid or colored solutions.
- High-precision dual-flow control system comprising a calibrated positive-displacement piston pump and ultra-stable electromagnetic flow regulator—ensuring volumetric accuracy better than ±1% and flow stability within ±0.5% over extended runs.
- Regulatory-grade software (V8.9 Integrated Edition) compliant with FDA 21 CFR Part 11, including five-tier user permission management, full electronic audit trail, digital signature capability, automatic data backup/recovery, and power-failure recovery protocol.
- 1000-channel high-resolution detection matrix supporting customizable binning (e.g., 4-, 8-, 16-, 32-, 64-, 128-, or full 1000-channel mode), enabling simultaneous compliance with multiple global standards such as ISO 21501-4, ISO 4406, NAS 1638, and ChP 0903.
- Integrated temperature-controlled fluid path (0–80 °C) and ambient operating range (−15–50 °C), suitable for cold-chain injectables, ophthalmic solutions, and high-viscosity suspensions.
- Touchscreen GUI with bilingual (English/Chinese) interface, intuitive workflow navigation, and automated calibration verification using NIST-traceable latex standards or certified reference materials per JJG 1061.
Sample Compatibility & Compliance
The PLD-601 is validated for use with a broad spectrum of pharmaceutical and biotechnological matrices, including but not limited to: sterile aqueous injection solutions, lyophilized powders reconstituted in WFI, concentrated injectables, ophthalmic preparations, vaccines, bulk active pharmaceutical ingredients (APIs), medical device extracts (e.g., IV sets, syringes, infusion filters), and packaging component leachates. It supports direct measurement of filter challenge studies—including terminal sterilizing filter integrity verification and microfiltration efficiency quantification per ASTM F838 and ISO 13408-2. All hardware and firmware meet GLP/GMP environmental and documentation requirements. Calibration and verification protocols align with ISO/IEC 17025-accredited laboratories, including the National Defense Science and Technology Commission First-Level Particle Metrology Station (No. 116) and Northwest Metrology & Testing Center of China Institute of Metrology.
Software & Data Management
The V8.9 analytical software suite provides full lifecycle data governance—from acquisition and real-time visualization to statistical reporting and long-term archival. Raw pulse data, histogram distributions, cumulative counts, and channel-specific concentration profiles are stored in encrypted, tamper-evident binary files with embedded metadata (operator ID, timestamp, instrument serial, calibration status). Reports export natively to PDF, CSV, and XML formats, and include configurable templates aligned with ChP, USP, EP, and ISO reporting conventions. Audit trails log all user actions—including parameter edits, result deletions, and report generation—with immutable timestamps and operator attribution. System validation documentation (IQ/OQ/PQ) is provided, and software upgrades follow formal change control procedures consistent with Annex 11 and ALCOA+ principles.
Applications
- Pharmaceutical QC: Routine batch release testing of injectables per ChP 0903 and USP ; particulate profiling of prefilled syringes and auto-injectors; raw material cleanliness screening.
- Medical Device Evaluation: Quantitative assessment of particulate shedding from IV tubing, connectors, and terminal filters; validation of filter retention efficiency at specified pore sizes (e.g., 0.22 µm, 0.45 µm).
- Water & Solvent Monitoring: Particulate contamination grading of WFI, purified water, and ultrapure water per USP , EP 2.2.43, and ISO 8573-4.
- Biomanufacturing Support: Monitoring of cell culture media, buffer solutions, and final formulation steps where subvisible particle load impacts product stability and immunogenicity.
- Cross-Industry Use: Semiconductor slurry purity verification (CMP), nanoparticle dispersion characterization in coatings and inks, and contaminant analysis in food-grade emulsions and cosmetic suspensions.
FAQ
Does the PLD-601 comply with FDA 21 CFR Part 11 requirements for electronic records and signatures?
Yes. The V8.9 software implements role-based access control, complete audit trail logging, electronic signature workflows, and secure data storage—all validated per Part 11 Annex A guidance.
Can the instrument be configured for non-standard particle size ranges beyond 1–500 µm?
Yes. Optional optical configurations support extended detection from 0.03 µm to 3000 µm, subject to sensor module selection and hydraulic path optimization.
Is third-party metrological calibration included with purchase?
Calibration certificates traceable to national standards (e.g., JJG 1061, ISO 21501-4) are supplied at shipment. Optional annual recalibration services are available via accredited partners including the Northwest Metrology & Testing Center.
What sample volume is required for a single test?
Standard assay uses 5–10 mL per run; minimum detectable volume is 1 mL with precision pump control ensuring repeatability at low volumes.
How does the system handle sample carryover between runs?
Automated rinse cycles operate at up to 500 mL/min with programmable duration and solvent selection, achieving <0.01% residual carryover verified per USP carryover validation protocols.