- All
- Favorite
- Popular
- Most rated
| Brand | KHB |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | Polaris c1000 |
| Price Range | USD 63,000 – 91,500 (based on EUR/USD exchange parity and typical export pricing) |
| Instrument Type | Mid-Speed Fully Automated Analyzer (400–1000 tests/hour) |
| Reagents | KHB-licensed reagents only |
| Sample Types | Serum, Plasma, Urine, Whole Blood, Cerebrospinal Fluid (CSF) |
| Throughput | Constant-speed 1000 tests/hour |
| ISE module | 600 tests/hour (optional) |
| Brand | KHB |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Polaris c2000 |
| Price Range | USD 95,000–136,000 (FOB) |
| Instrument Type | High-throughput Fully Automated Analyzer (1000–2000 tests/hour) |
| Reagents | KHB-licensed in vitro diagnostic reagents only |
| Sample Types | Serum, Plasma, Urine, Whole Blood, Cerebrospinal Fluid (CSF) |
| Throughput | Constant-speed mode: 2000 tests/hour |
| Stat Emergency (SE) mode | 600 tests/hour (optional) |
| Brand | Kehua Bio (KHB) |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic (China) |
| Model | ZY-1200 |
| Instrument Type | High-throughput Fully Automated Analyzer (1000–2000 tests/hour) |
| Assay Speed | 800 photometric tests/hour (biochemistry), 400 tests/hour (optional ISE module) |
| Minimum Sample Volume | 1.0 µL (0.1 µL increment) |
| Minimum Reagent Volume | 10 µL |
| Minimum Reaction Volume | 90 µL |
| Sample Types | Serum, plasma, whole blood, urine (per reagent package insert) |
| Reagent Compatibility | Kehua Bio–branded IVD reagents only |
| Regulatory Status | NMPA-certified Class II IVD device for clinical use in China |
| Test Menu | ~80 assays available with CE-IVD–marked or NMPA-registered reagents |
| Brand | Kehua Bio (KHB) |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | ZY-680 |
| Instrument Class | Mid-Speed Fully Automated Analyzer (400–1,000 tests/h) |
| Assay Throughput | 680 photometric tests/h (biochemistry), 400 tests/h (ISE module, optional) |
| Sample Types | Serum, plasma, whole blood, urine (per reagent package insert) |
| Reagent Compatibility | Kehua Bio–branded reagents only |
| Wavelength Range | 340–800 nm |
| Optical System | Holographic flat-field concave grating + diode array transient detection |
| Temperature Control | Water-bath incubation at 37.0 ± 0.1 °C with real-time display and out-of-range alarm |
| Cleaning System | Multi-step warm-water cuvette washing, independent polished vortex stirrer, KHB-specific cleaning solution |
| Sample/Reagent Capacity | High-capacity sample and reagent carousels with priority emergency sample insertion, auto-dilution & retest |
| Reagent Cooling | Peltier-based refrigeration (2–8 °C) |
| Reagent Monitoring | Real-time residual volume tracking with test-count estimation |
| Cuvette Design | Optimized geometry with bottom-structured reagent vials for minimized dead volume |
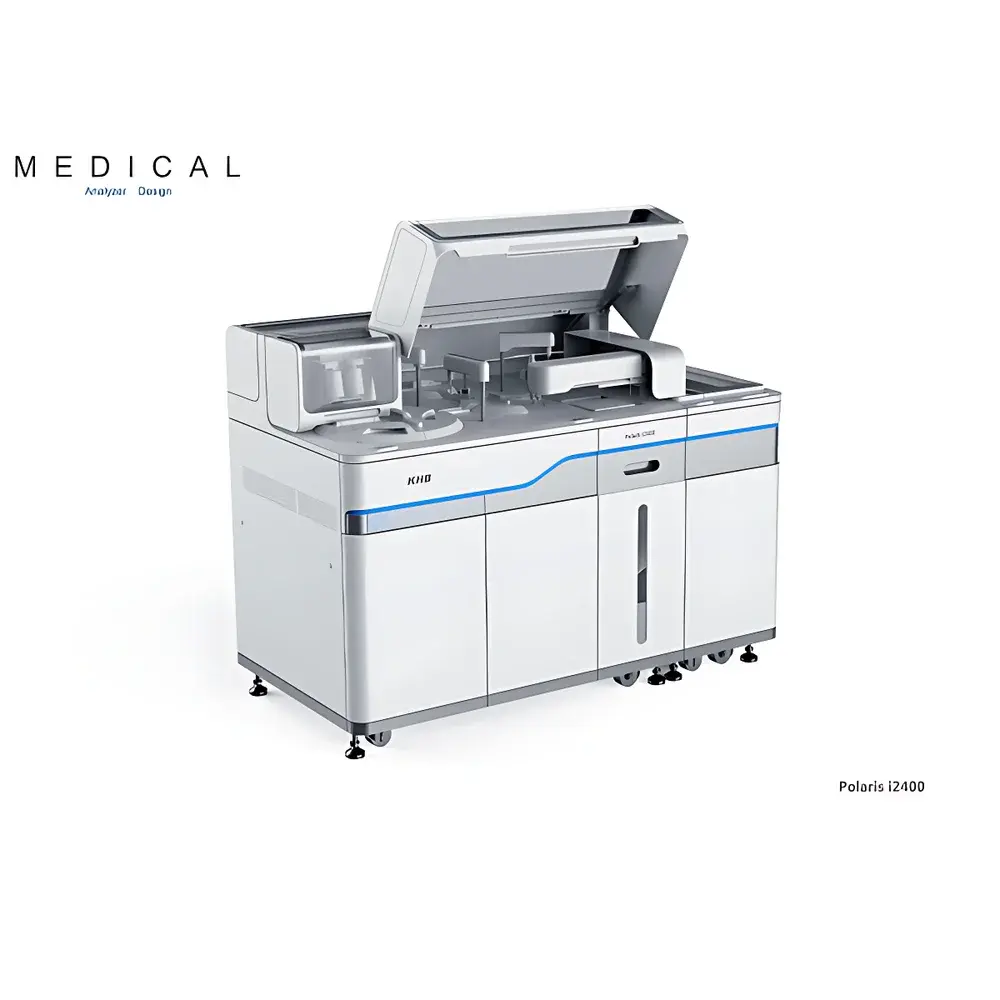
| Brand | KHB |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Polaris i2400 |
| Price Range | USD 28,000 – 70,000 (FOB Shanghai) |
| Assay Principle | Indirect Chemiluminescence |
| Automation Level | Fully Automated |
| Throughput | 240 tests/hour |
| Sample Types | Serum, plasma, and other liquid clinical specimens |
| Detection System | ALP-AMPPD chemiluminescent substrate system |
| Sensitivity | ≤10⁻²¹ mol/L |
| Temperature Control Accuracy | ±0.1°C |
| Sample Carryover Prevention | Disposable tip/cup system |
| Regulatory Compliance | CE-IVD, NMPA Registration Class II/III (as per test menu), ISO 13485 certified manufacturing |
| Brand | Kehua Bio (KHB) |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | mini LAS |
| Pricing | Upon Request |
| Footprint | 6 m² |
| Throughput | Up to 2000 tests/h (clinical chemistry), Up to 240 tests/h (immunoassay) |
| Sample Loading Options | Gravitational pouring, Rack-based loading, Pneumatic tube transport |
| Sample Processing | Integrated pre-analytical and post-analytical module with automatic cap piercing and re-capping |
| Quality Control | Scheduled auto-start, remote activation via WeChat mini-program, AI-powered QC flagging logic |
| Sample Integrity | AI vision-based sample inspection (clot detection, hemolysis, lipemia, volume insufficiency) |
| Carryover Prevention | Dual-wash probe system with real-time monitoring, compliant with CLSI EP26-A guidelines |
| Regulatory Compliance | Meets GB/T 26124–2010 (Clinical Chemistry Analyzers), YY/T 1255–2015 (Immunoassay Systems), ISO 15189 pre-analytical requirements |
| Brand | KHB |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic (China) |
| Model | Polaris i1800 |
| Automation Level | Fully Automated |
| Throughput | 180 tests/hour |
| Sample Types | Serum, Plasma |
| Reagent Cartridge Compatibility | KHB Polaris® i2400 |
| Detection Principle | ALP-AMPPD Enzyme-Enhanced Chemiluminescence |
| Sample Volume Precision | CV ≤ 2% at 10 µL, CV ≤ 1% at 200 µL |
| Sample Capacity | 60 positions (6 racks), barcode-readable |
| Reagent Positions | 20, refrigerated (2–8 °C), with magnetic bead reagent agitation |
| Reaction Cup Capacity | 500 disposable cups, auto-replenishable |
| Substrate Delivery | Dual-channel, hot-swap capable |
| Waste Management | 500-cup waste bin, single integrated waste liquid reservoir |
| Brand | Kehua Bio (KHB) |
|---|---|
| Origin | Shanghai, China |
| Model | ZY-400 |
| Instrument Type | Low-speed fully automated analyzer (0–400 tests/h) |
| Sample Types | Whole blood, serum, plasma, urine (per reagent instructions) |
| Throughput | 400 photometric tests/h (biochemistry) |
| Cross-contamination rate | <0.08% |
| Sample volume range | 1.5–70 µL (0.1 µL increment) |
| Cuvette material | Permanent optical glass |
| Reaction temperature control | Dual-phase liquid-gas thermostatic system, maintained at 37 ± 0.1 °C with real-time display and over-limit alarm |
| Calibration methods | Linear (K-factor, 2-point/multi-point), nonlinear (spline, piecewise linear, exponential, logarithmic, 3P/4P/5P logistic) |
| Analytical methods | End-point (single/dual), kinetic (rate), fixed-time, ISE potentiometry |
| Reagent refrigeration | Semiconductor-cooled reagent tray |
| Reagent monitoring | Real-time residual volume tracking with estimated remaining test count |
| Interface | Bidirectional LIS/HIS connectivity via standard network protocols (TCP/IP, ASTM, HL7) |
| User management | Role-based access control with audit trail |
| Compliance | CNAS-accredited medical laboratory validation framework |
| Power management | 24-hour continuous operation with auto-sleep/wake cycle |
Show next